Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
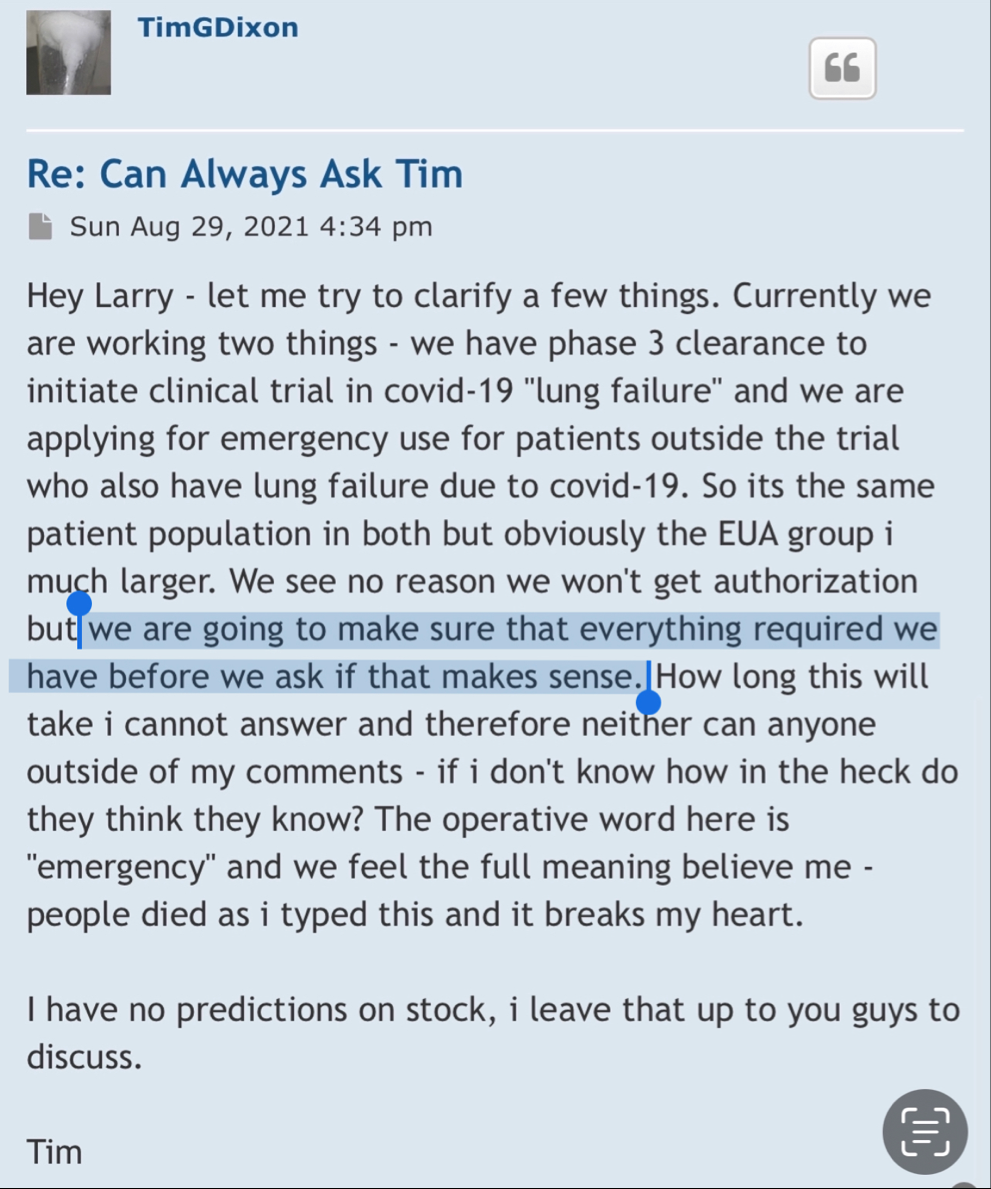
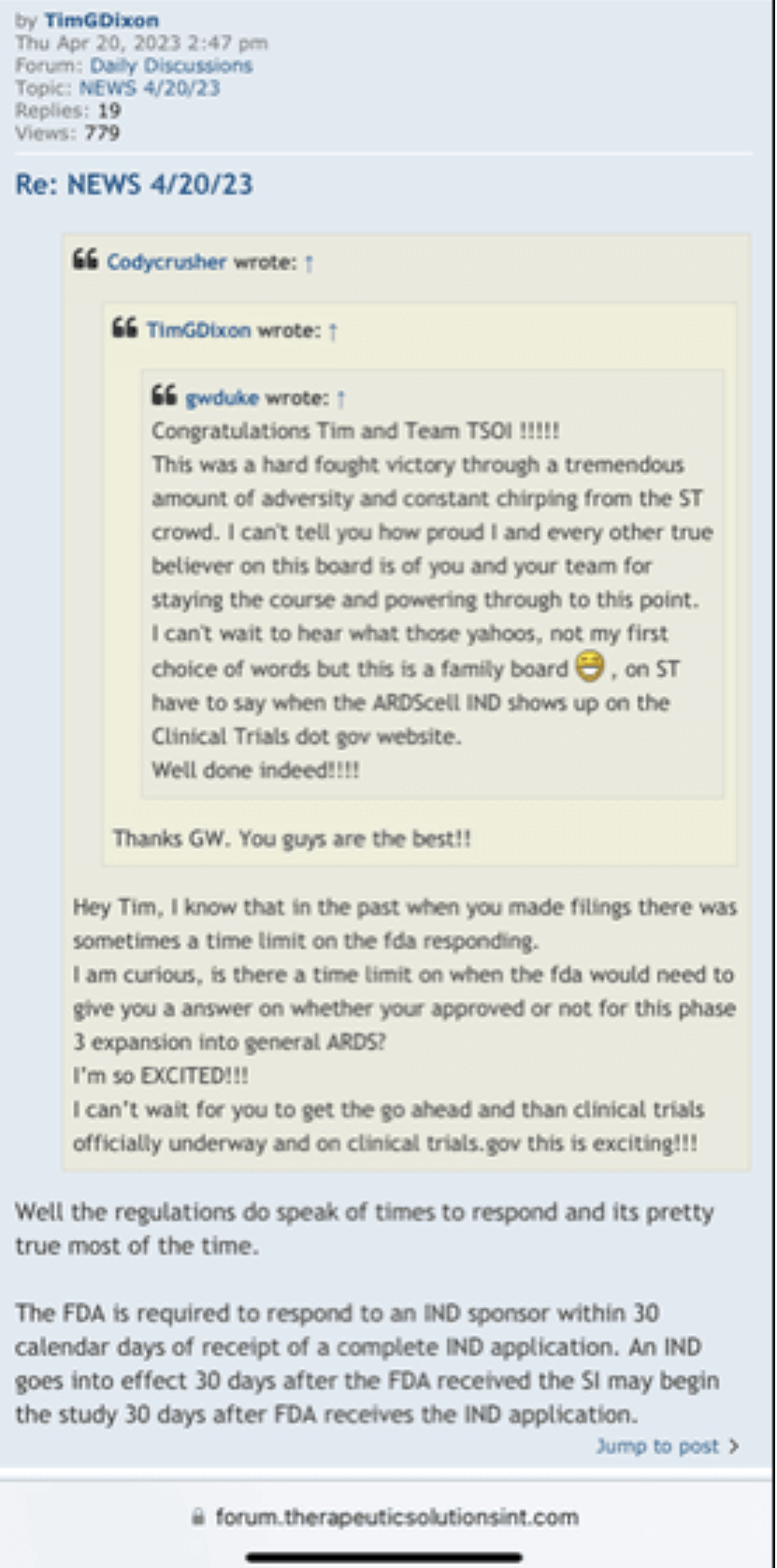
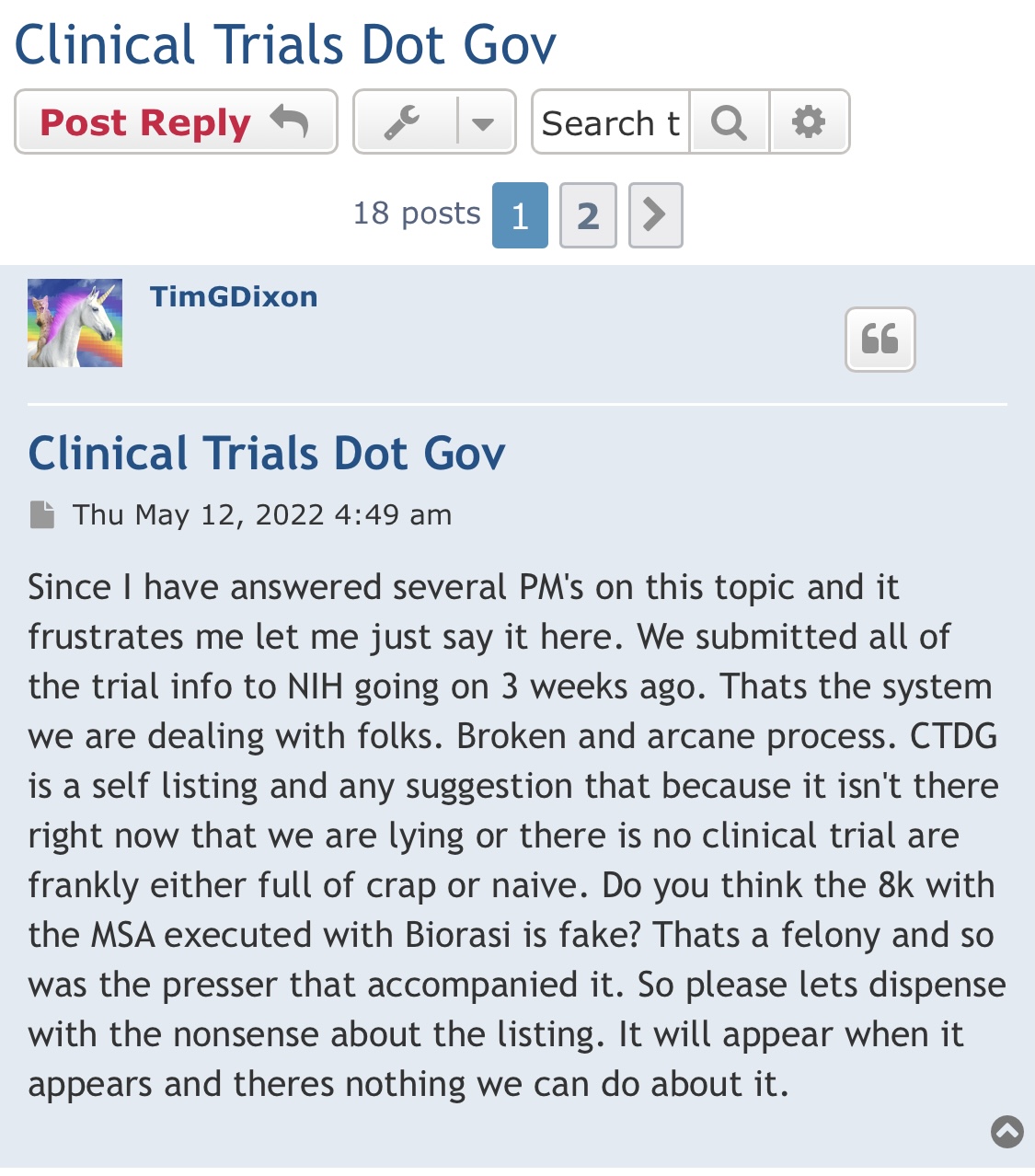
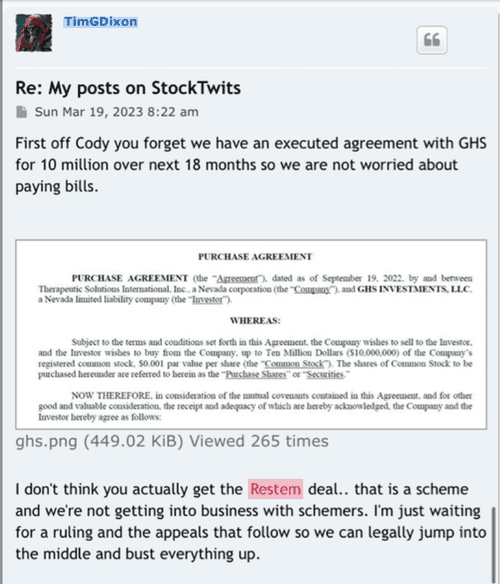
Divi letters went out yesterday this is from the company.
I see! So if Kooscous is a Big Pig Loser, because everything he's done all his miserable life has been a series of failures and swindles and this is how you have always perceived him, explain why you would of ever became a shareholder in his company? Remember you used the phrase ”all his miserable life” which would mean you would be going back to the beginning on his entire working career to review his past achievements! Now let’s get to what is more important on why you are posting others posters TSOI posts word for word knowing you have NEVER posted on this TSOI board ever before as seen in the bottom two links provided? Strangely enough the person who originally wrote those posts is now commenting on YOUR post here without having the ability to identify the information you are providing was actually written by himself? Hmmm???
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173590560
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174160438
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174154637
Yes, we shall see, but to be clear I am not making any evaluations of the science. I am saying that:
1) TSOI execs lied to shareholders
2) Science is evaluated via replicable, controlled studies and commercialization is achieved via the clinical trial process. TSOI has made little to no headway in the latter case and likely remains years from commercialization and unlikely to get there (as is case with any early bio that has never completed a clinical trial and is millions in debt).
3) TSOI is heavily diluting and no one in the space appears interested in its IP or current products (sales are woeful and 20% discounted shares dumped via GHS is best "funding" it has been able to come up with)
You are right about shareholders crushed, but in the end I think you will be wrong about the science. But we will see...
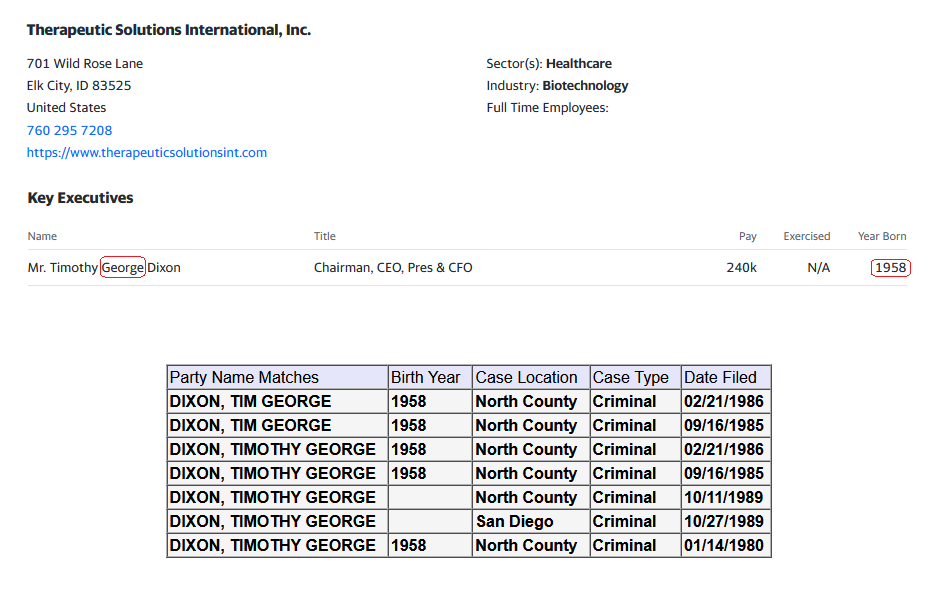
Superior Court of California/San Diego County
https://roa.sdcourt.ca.gov/roa/faces/CaseSearch.xhtml

Do you know the location of the case? (State/County/District)?
We will see where that is at in 30 days.
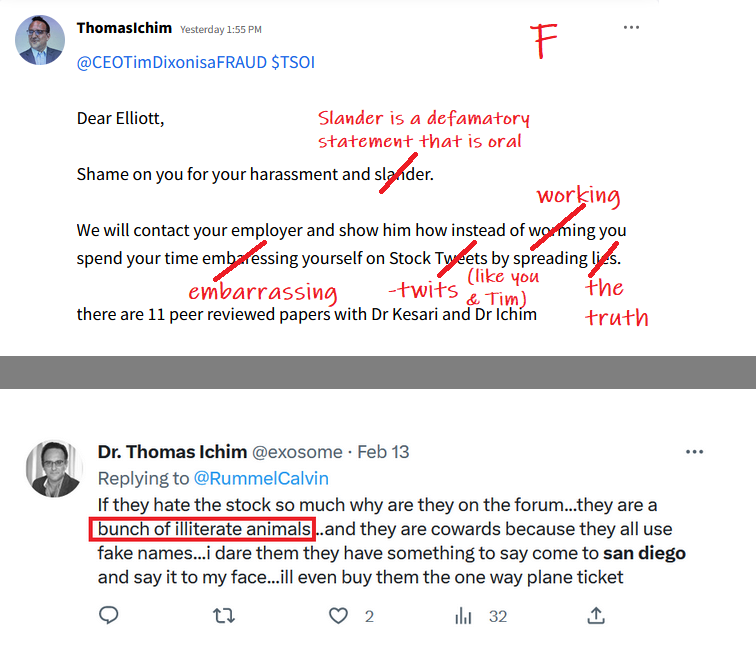
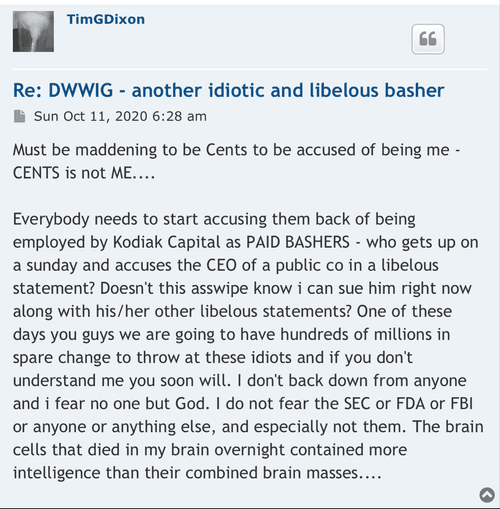
UMMMM ***INVESTORS ALERT**** TSOI Corp, Thomas Ichim and Dr. James Veltmeyer are being sued for FRAUD, breach of Fiduciary Duty;
- Conversion;
-Accounting;
- Imposition of a Constructive
Trust; and
-Intentional Interference with
Contractual Relations
https://stocktwits.com/rustymaverick/message/568385484
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174129008&txt2find=RyRy
can you point out where its wrong? I mean come on
When what is presented can't be refuted
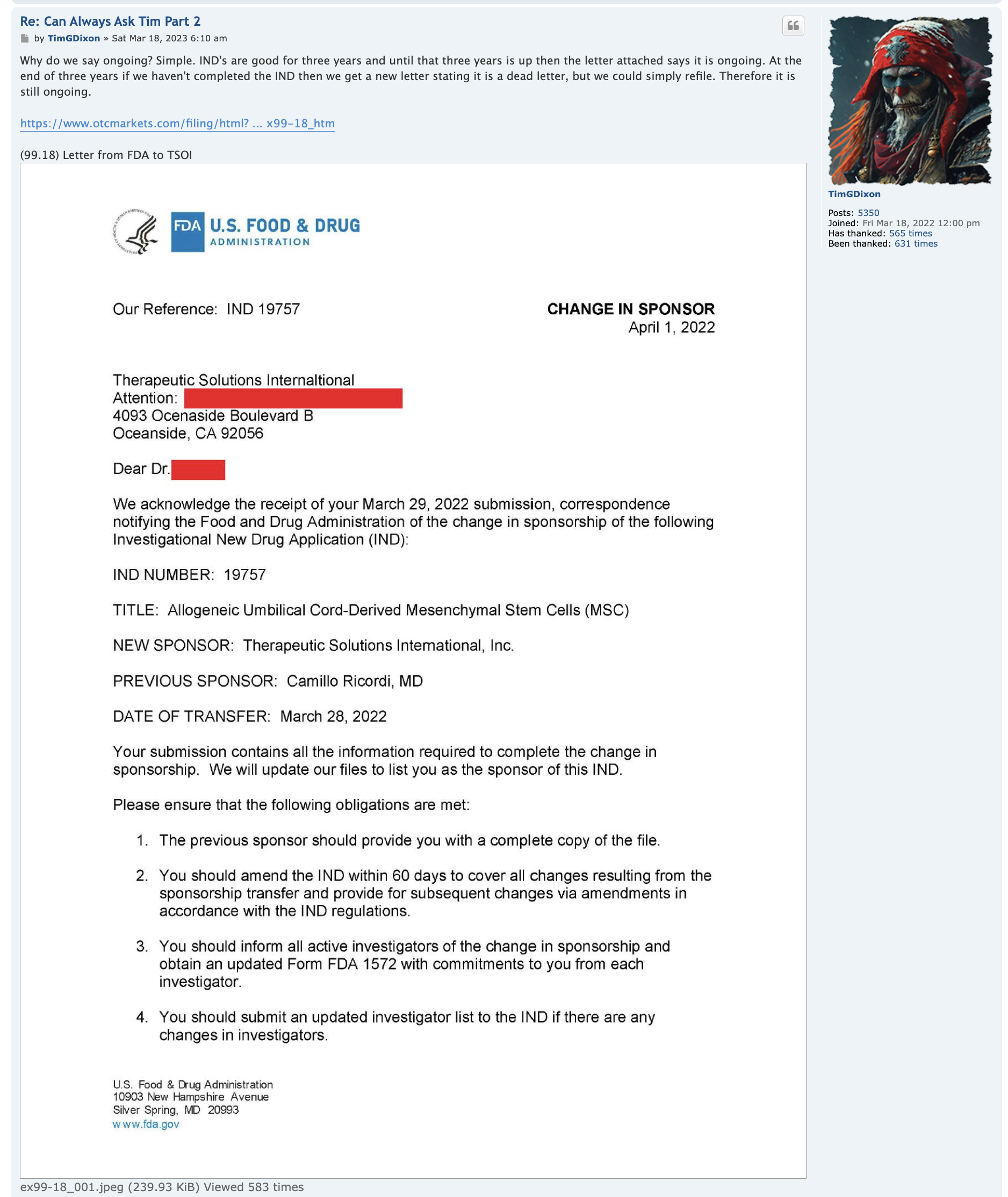
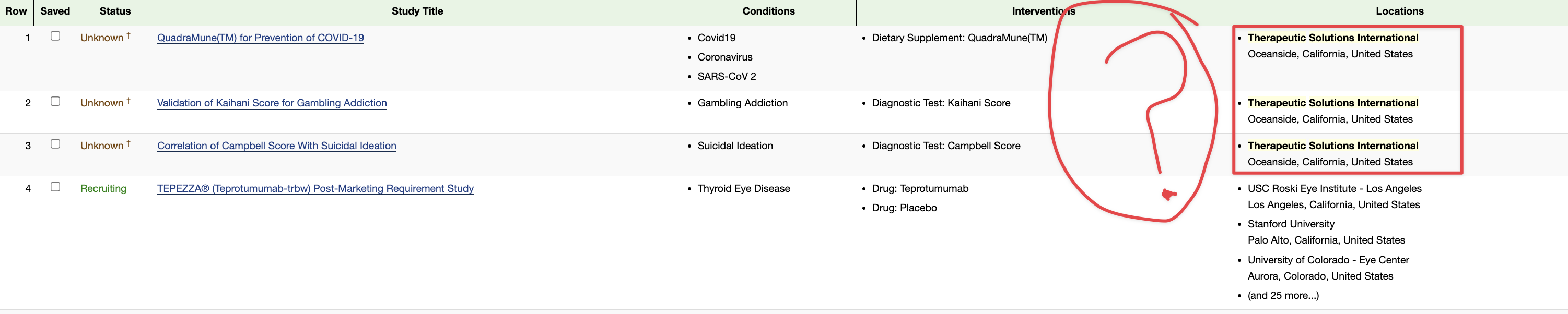
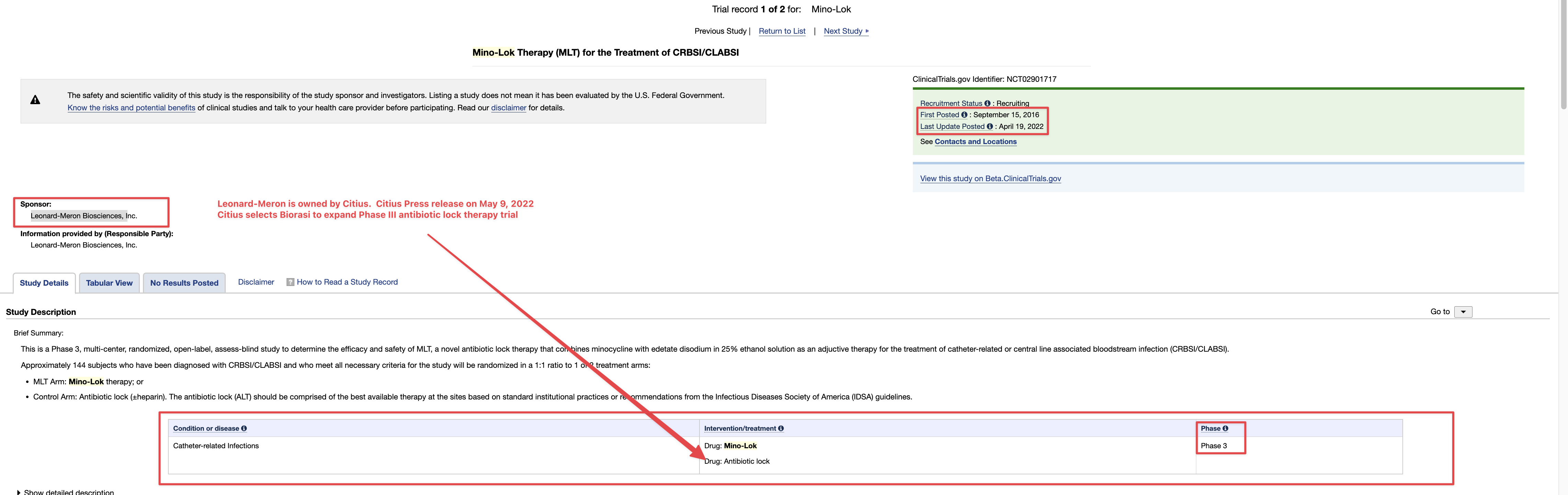
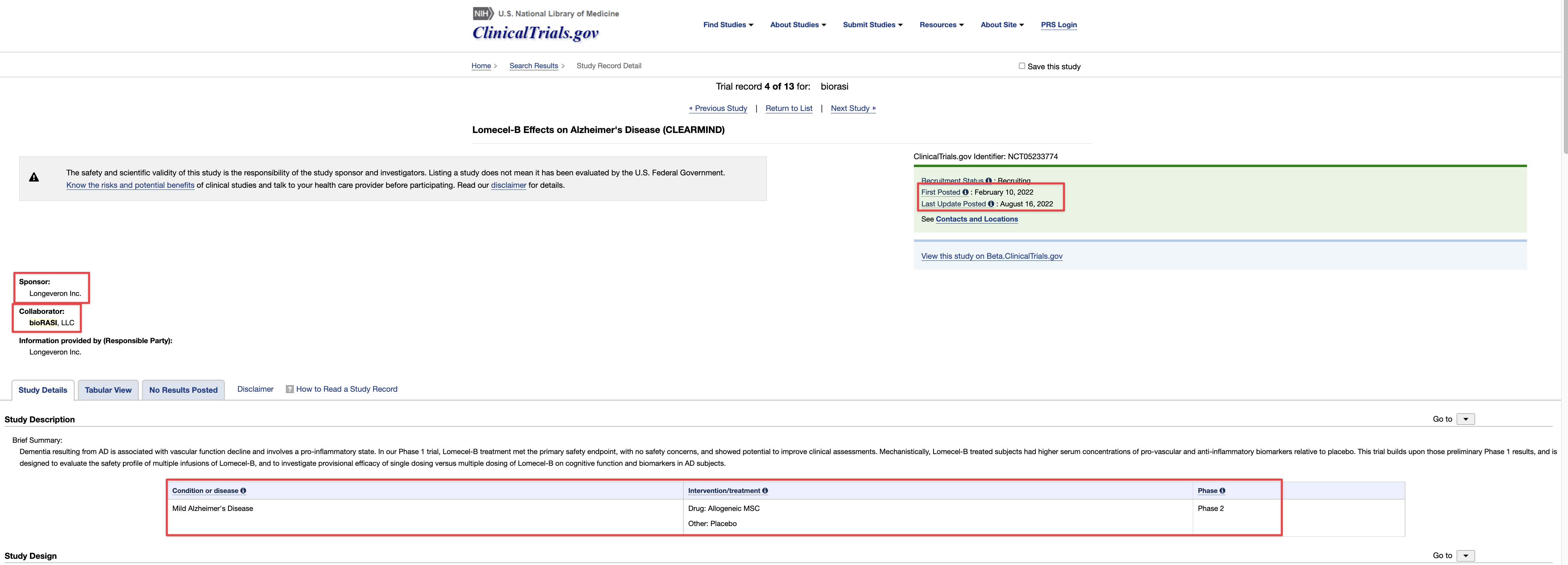
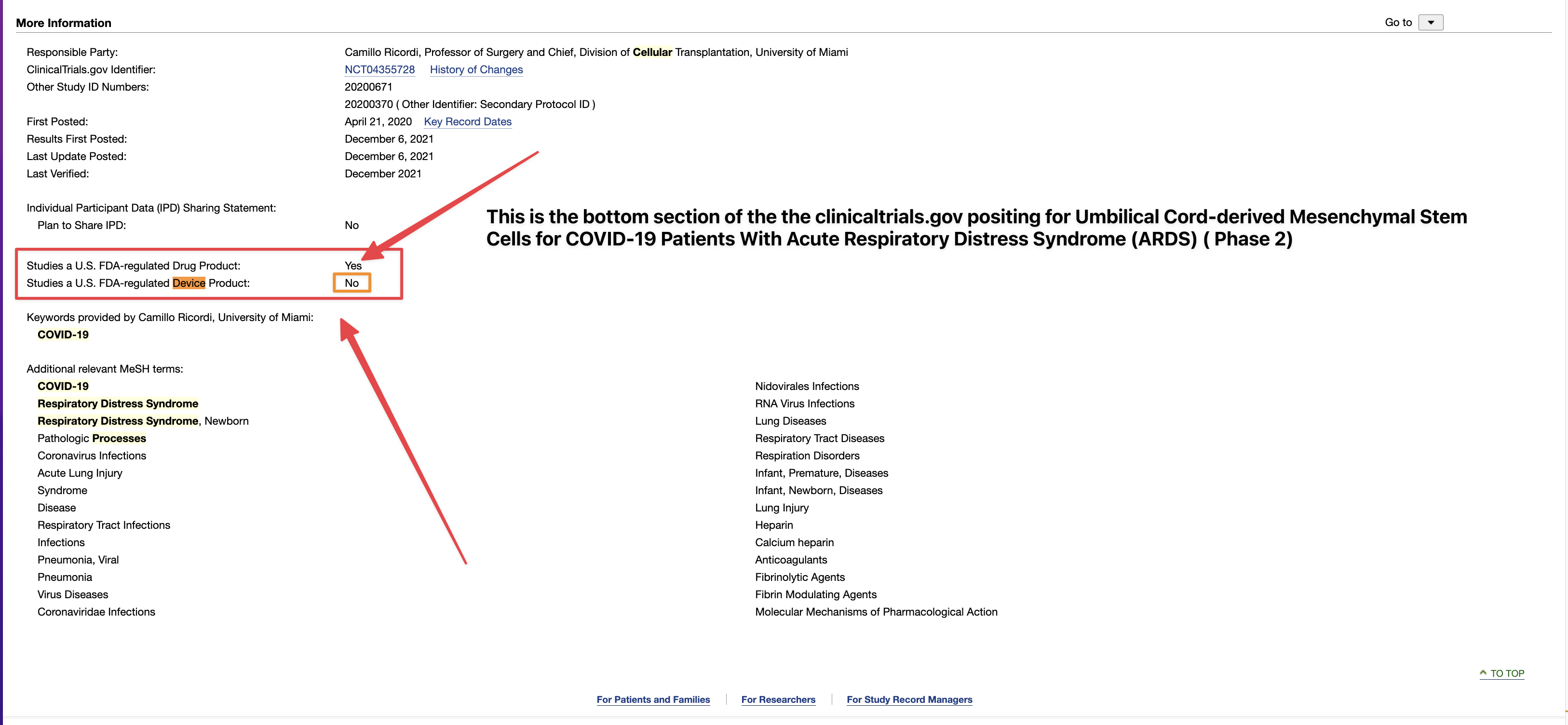
Ongoing means, with respect to a clinical trial of a drug product (including a biological product) or a device product and to a date, that one or more human subjects is enrolled in the clinical trial, and the date is before the primary completion date of the clinical trial.
ClinicalTrials.gov is a registry and results database of privately and publicly funded clinical studies conducted around the world. The resource is provided by the U.S. National Library of Medicine. Each study record includes a summary of the study protocol.
NIH and ICMJE Reporting Requirements. NIH requires all clinical trials, regardless of study phase, to register on ClinicalTrials.gov.
no sell no buy now
Any guesses, as to the name of the guy at the reins?
https://www.shutterstock.com/image-illustration/donkey-running-after-carrot-518056597
Everyone...please heed the info...It's BOLD and RED! LOL
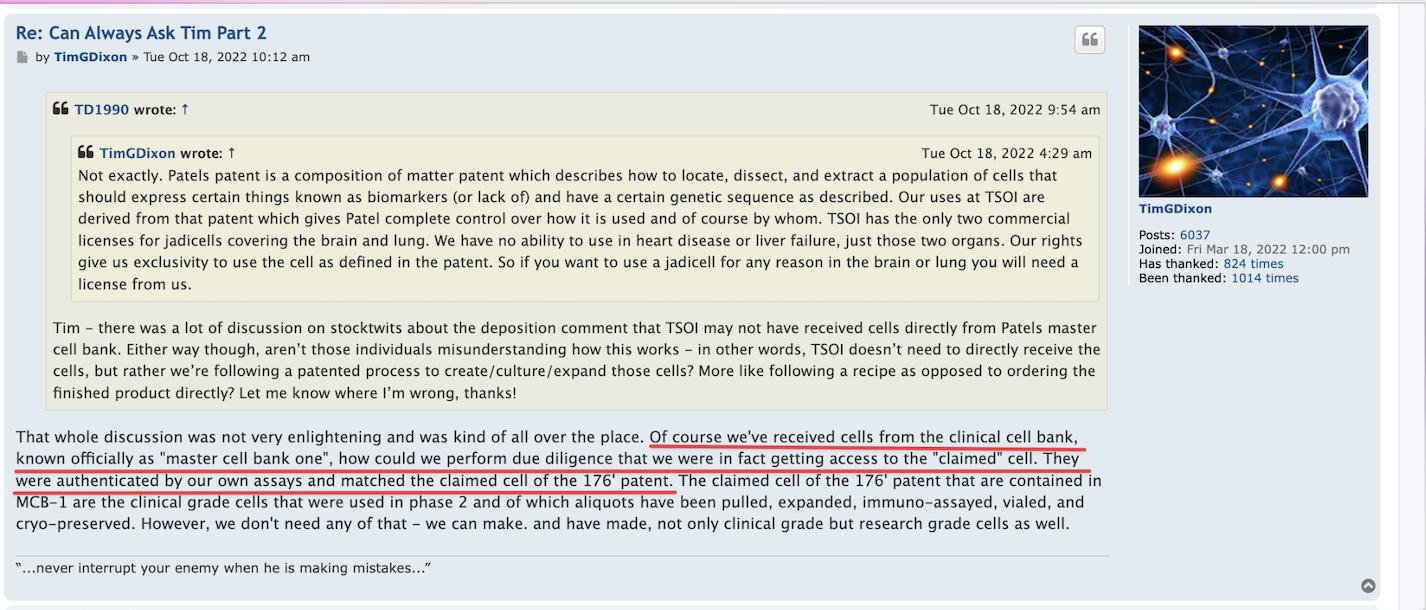
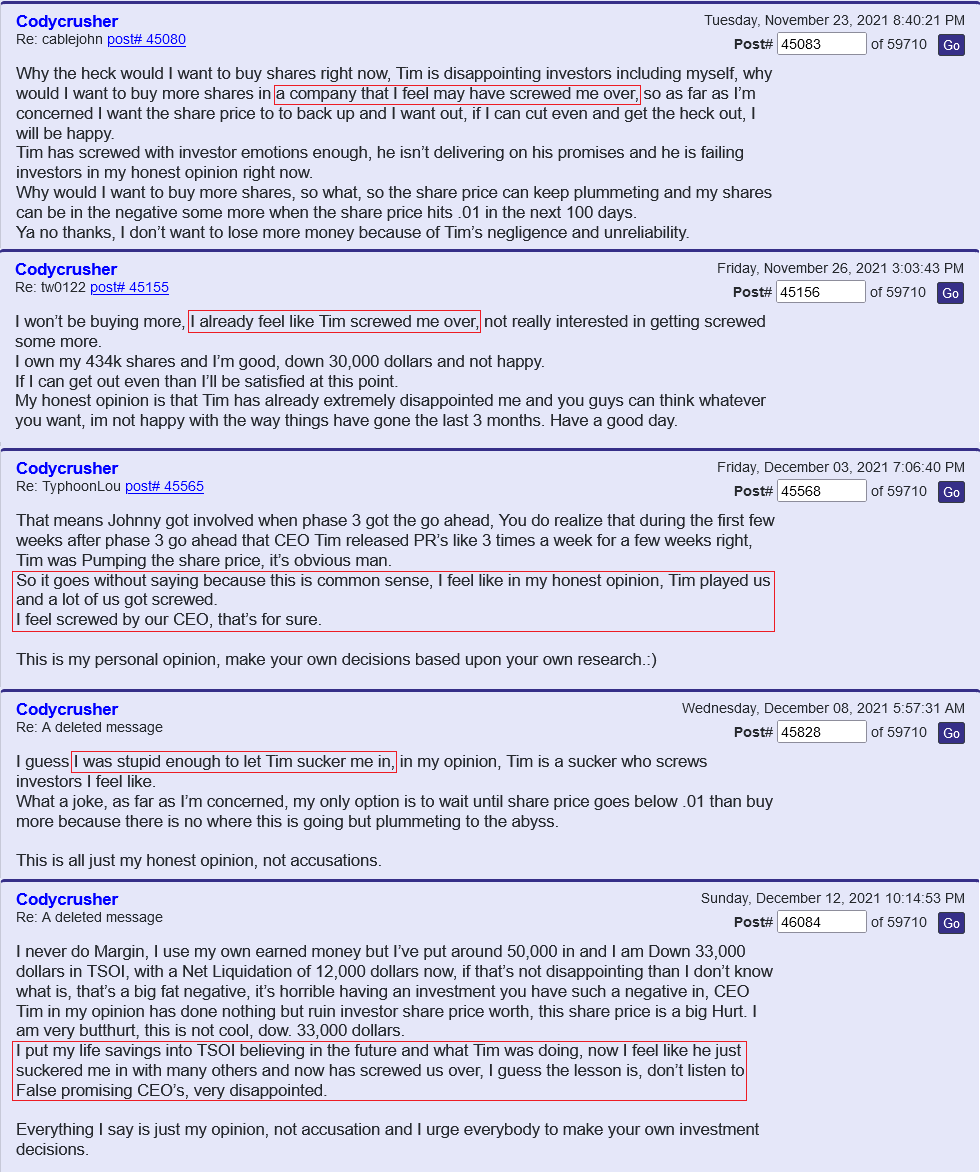
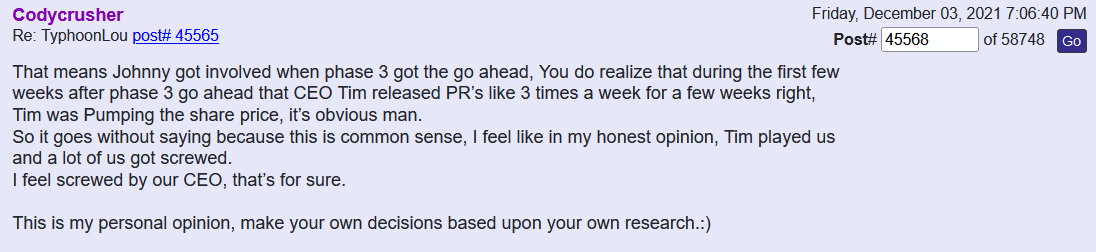
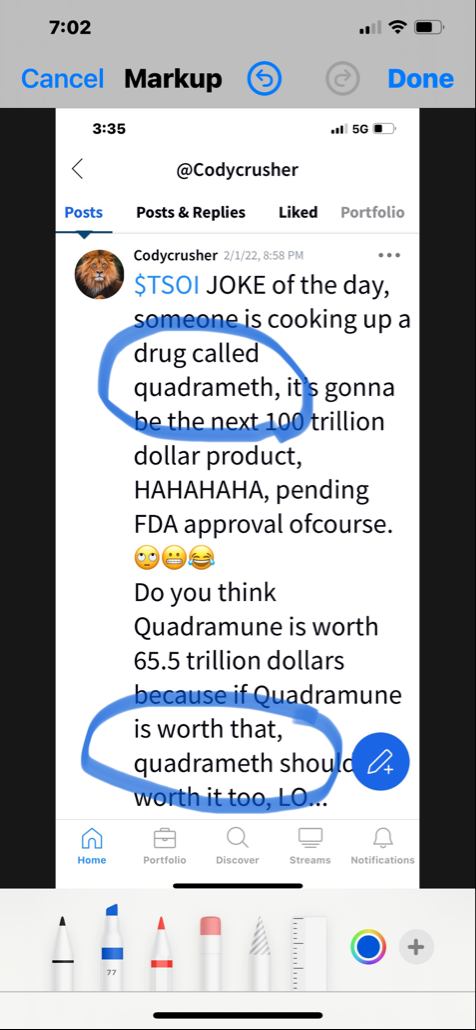
Define crook. We know that Ichim has had journal articles retracted for ethical issues and 'fictional science'. We also know that he lied to TSOI investors about an ongoing P3 trial for months. Furthermore, Ichim has been associated with many more failures and companies that have hurt investors (Batu, RGBP, CELZ, TSOI) than he has "successful business ventures." Those others are not all that important to me. TSOI longs have lost up to 99%+ and ~70% over past year alone.
Real science is established via peer-reviewed/replicable clinical trials. TSOI has never completed one of those. What do you know about the legitimacy of that "science." More importantly, stocks are about business and shareholder value first and foremost, not ill-supported science. That science is years away from commercialization and unlikely to get there. Sales are declining, revenue is pathetic and massive dilution remains the primary funding source. No one else seems interested in partnering with nor funding TSOI. I wonder why. The best science is unlikely to over come all that.
People can talk about future projections and rewards all they like. The fact is TSOI investors have lost BIGLY to date. The FDA does not have any aging requirements and TSOI is diluting like crazy.
Cheers.
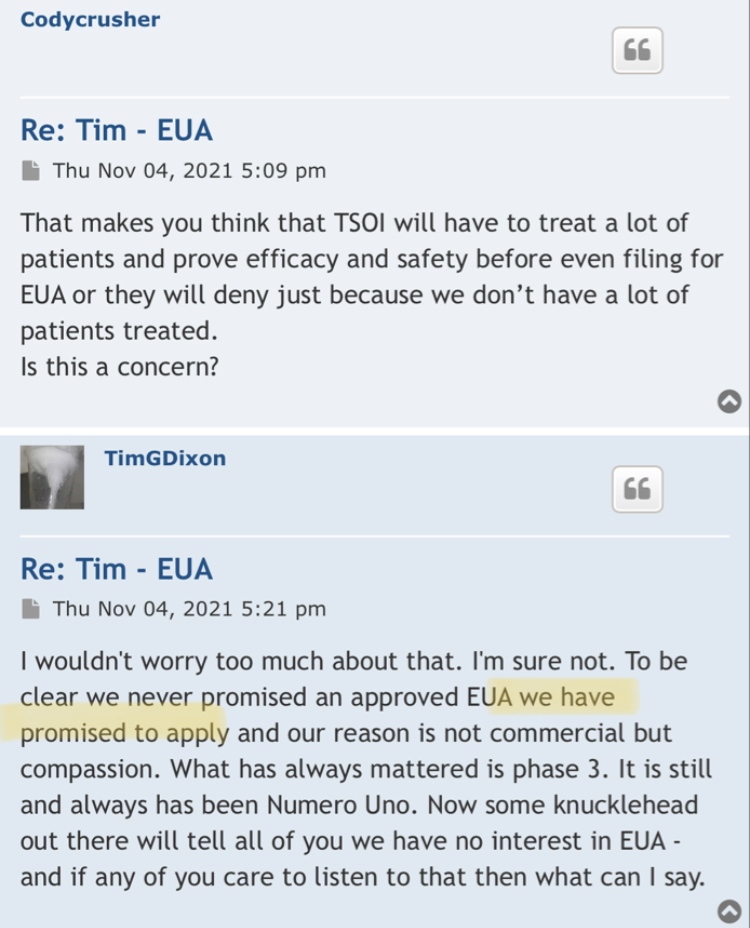
Please, Tom is not a crook. He has been successful in past business ventures.
When business plans stall, move sideways, are delayed - it is so easy to throw out the word "Scam".
But real business goes through ups and downs, and many don't make it - many times where plans didn't work out, not because everyone is a crook. That is a simpletons way of looking at everything.
I don't know if TSO will succeed, but I think those who persevere with TSOI will ultimately be rewarded. There is real science behind the fields they are in. If they get their cells okayed by FDA (aging), clinical trials may start in fall / winter of 2024. Even if just 20% of success, it is worth it down at these levels.
Warning !!!
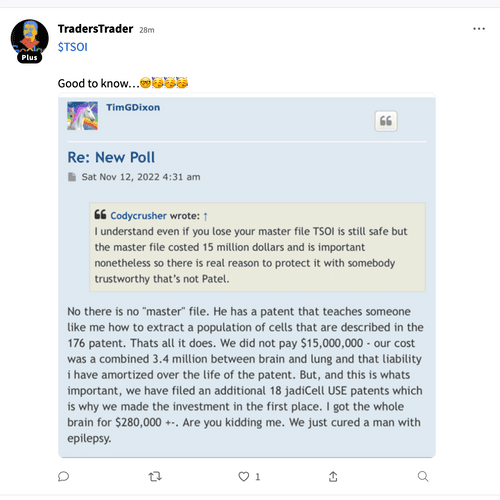
Thomas Ichim who produced more than 80% of RGBP's "patents" turns out to be a crook just like the CEO of Regen Biopharma the lying crook David Koos.
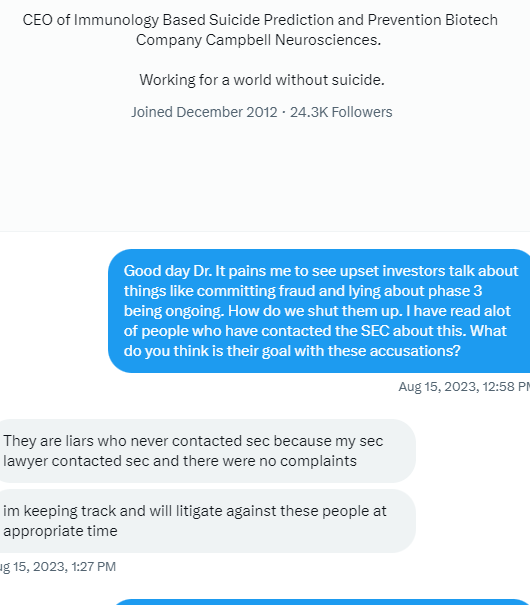
$*SOI ***ATTENTION ALL INVESTORS WHO WERE SCAMMED BY CEO TIM DIXON and Thomas Ichim**** As most of you know we have been scammed by CEO Tim Dixon. I would encourage you all to submit a "tip" with the SEC and spread the word. Do not let this crock of turd CEO get away with this crap. It's time to make him pay as we all have believed his lie after lie after lie. Here is the link which I will post daily. Its time. https://www.sec.gov/whistleblower/submit-a-tip This is one way to possibly get paid back by the SEC as a whistleblower!
**FRAUD ALERT**** as most with any sort of common cents knows this is a scam. Has been for well over a decade. Lie upon lie the only real truth is this is a share selling operation run by career criminals CEO Tim Dixon and Thomas Ichim a crackhead. Continue to report their mangy asses to the SEC. I mean...... those are the actual facts.
$TSOI is understandably around historic lows and 99% loss of "value"
💩 Leadership is dishonest + extremely unprofessional scumbags
💩 IP is being diluted/sold for peanuts across 8 SCAM Subsidiaries
💩 NO clinical trials registered as current (15+ years in existence)
💩 Very little cash on hand
💩 No FDA Phase III IND (couldn't afford it)
💩 Grossly limited funding (stock given away at 20% discount)
💩 Substantial/ongoing dilution
💩 Uses RTT to cloak chronic incompetence & BS while touting lifesaving work. Yet they can't get an actual clinical study registered to save their own life.
💩 Homophobic, narcissistic CEO calls shareholders "pigs" and mocks dead mothers
💩 Only 4 granted patents (most of which are related to blueberry/broccoli pills with crappy sales)
💩 Pathetic/decreasing revenue
💩 Beaver-infested company land (that
CEO squats on)
💩 Dumbest gaggle of paid pumper clowns on ST's
💩 Insiders never buy shares
💩 Stuck in lawsuits
You gotta do something though if you are seeking to dilute billions of shares at as high a pps as possible??? What a great philosophy you have to remain a dedicated TSOI shareholder!!! The vast majority of investors surely wouldn’t tolerate such actions by the company they’re invested in if that is what they truly believed and simply separated themselves from the situation!!!
Yes it is. Especially considering how illusory many of the carrots have proven to be.
You gotta do something though if you are seeking to dilute billions of shares at as high a pps as possible.
The number of carrots this company dangles is astounding.
Just the facts!!! Dixon is doing al he can to sucker in more investors!!
https://stocktwits.com/rustymaverick/message/573207781
🤣 Oh that is all you have? I thought there might be something more reliable you were basing your claims on.
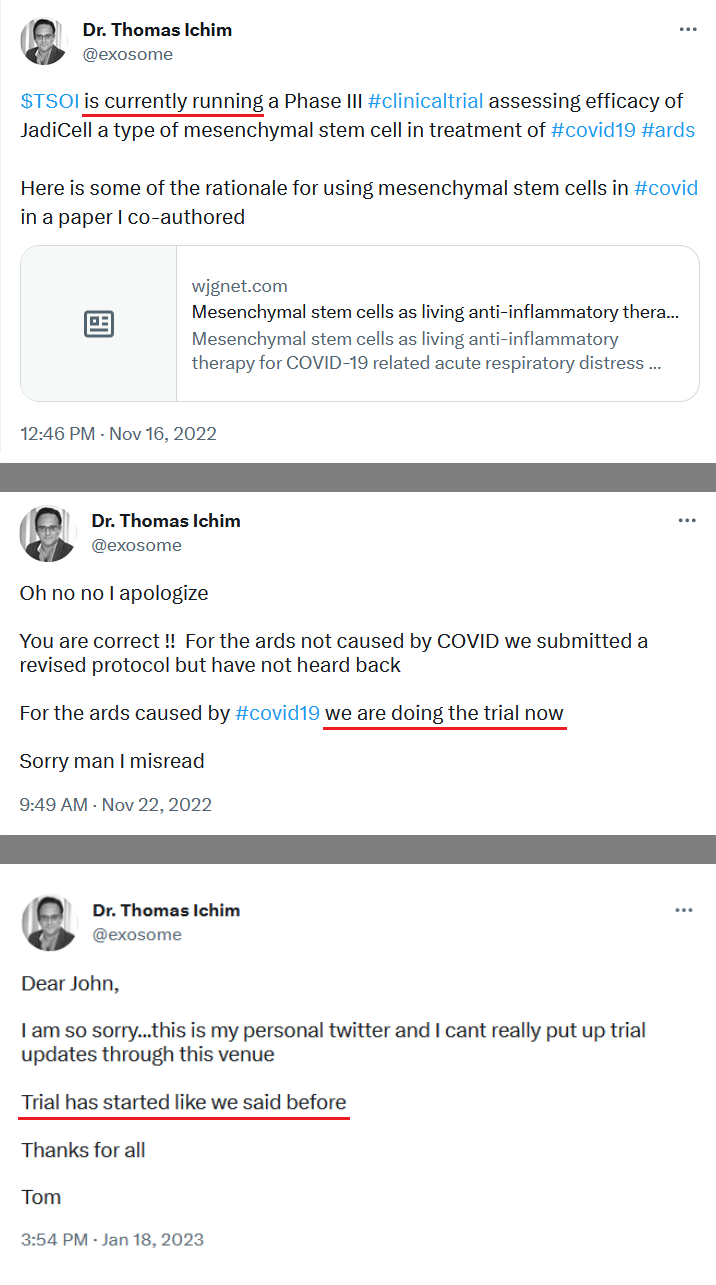
That paragraph from the press release (issued by a company that has lied in previous press releases or at the very least incompetently provided false information in press releases) reads:
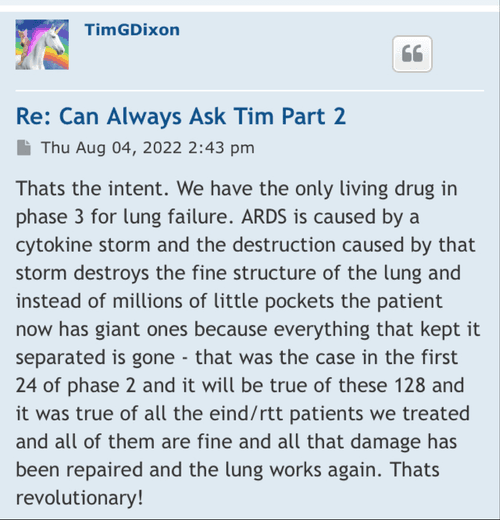
"Therapeutic Solutions International owns a Phase III Investigational New Drug application for treatment of COVID-19. However, the Company is working on a new application for treating “all-cause” acute respiratory distress syndrome (ARDS) using the company’s patented JadiCell1, a true mesenchymal stem cell. Additionally, the Company has filed a Phase I/II clinical trial for COPD and possesses preclinical data for pulmonary fibrosis. All of these conditions are examples of lung inflammation."
We know that TSOI does not own that IND. It defaulted on the 1.8 Million that was due in 3.23. We also know that the new application re: ARDS was filed over a year ago and was likely not approved. Why would FDA approve it when TSOI does not have a completed P2? 🤣🤣🤣 Given all of that BS why would anyone think the PI/II application is true?
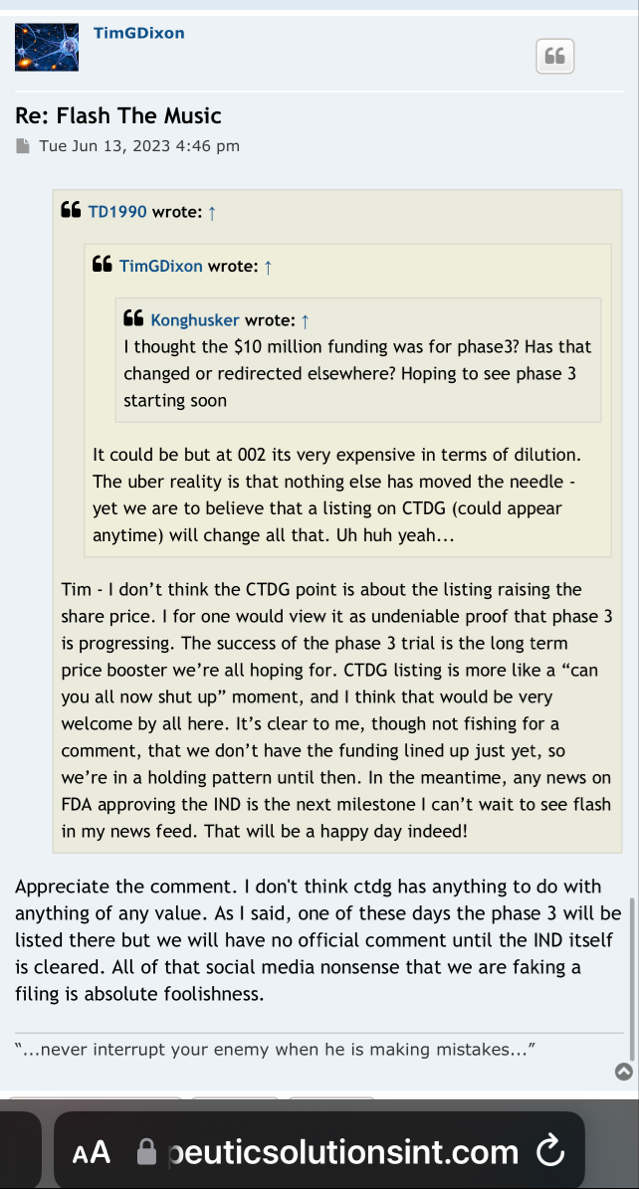
We will see where that is at in 30 days. That one is the only one that actually seems to have a chance. TSOI will be starting by trying to complete a registered P1 for a change, if and when it gets the opportunity and funding to actually do so There is no P3 unless you believe crazy stories make it real.
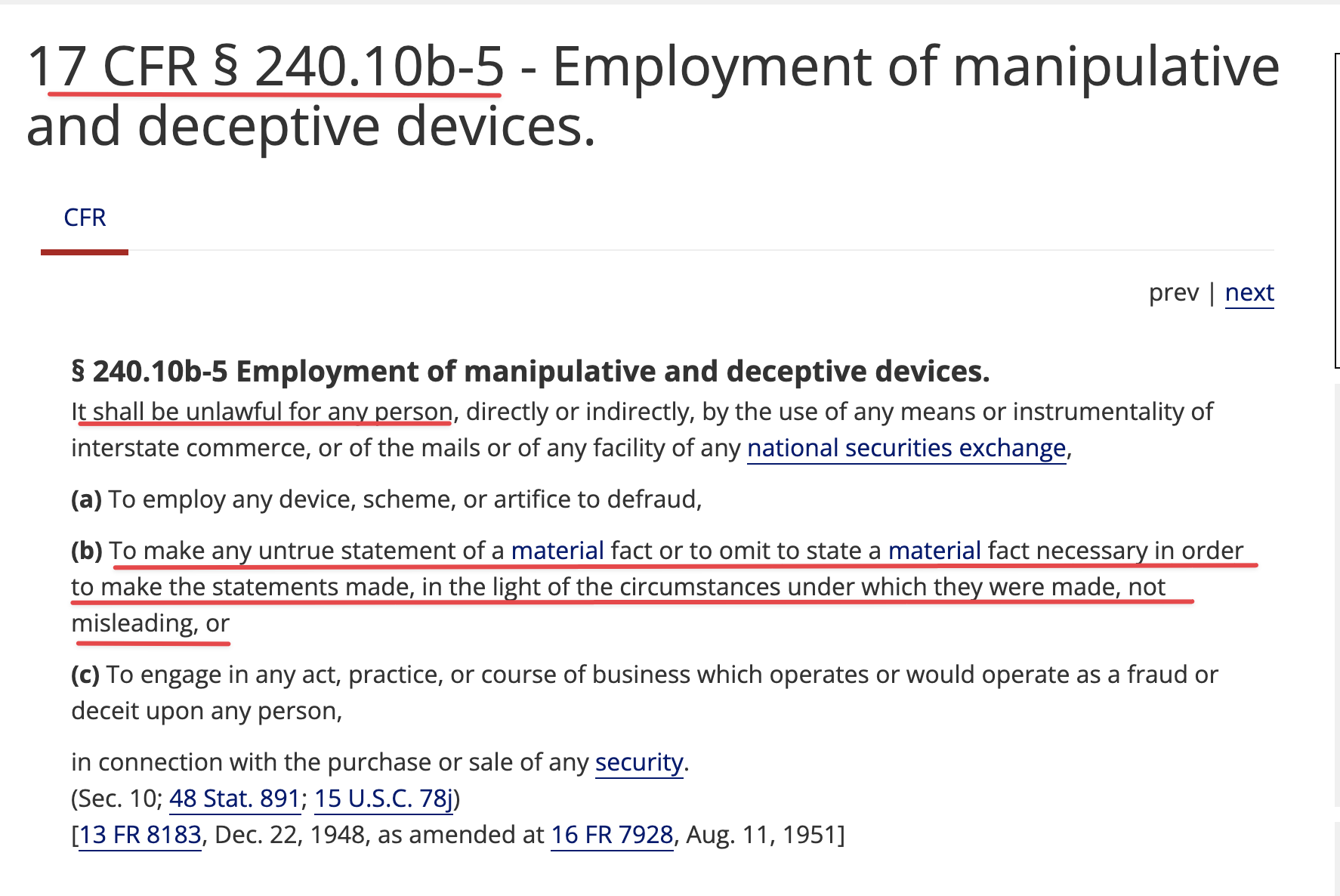
Again you can't refute my point (yet your provided info seems to validate it) as Patents & Registered Clinical Trials are not the same ![]()
USPTO & http://clinicaltrials.gov
Contract Default means the failure by a party to a contractual agreement to perform or meet contract terms and conditions.
If you have received the NOA, congratulations! As you approach the end of the journey for this particular patent application, you’ll need to take certain final steps to wrap up your patent.
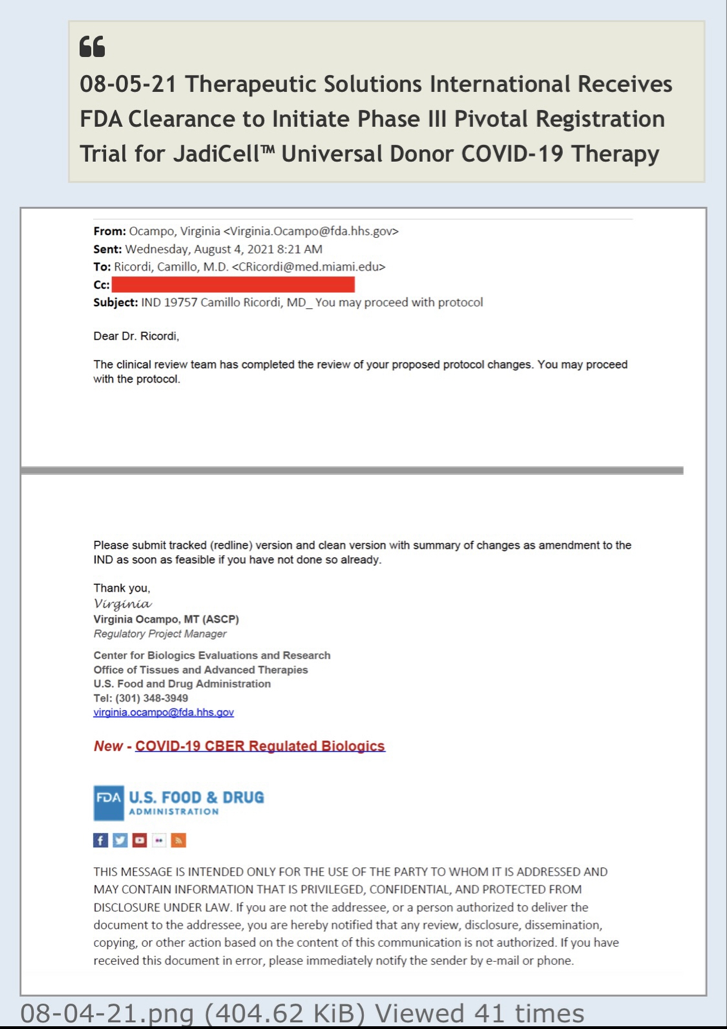
Therapeutic Solutions International owns a Phase III Investigational New Drug
True, we're in the drums & trumpets stage of the patent process.
What is a Notice of Allowance in a patent application?
A Notice of Allowance (NOA) is a USPTO document indicating that a patent application has been allowed. The approval of your patent application is basically the finish line of your journey and presumably the goal of every applicant. If you have received the NOA, congratulations! As you approach the end of the journey for this particular patent application, you’ll need to take certain final steps to wrap up your patent.
USPTO & ClinicalTrials.gov http://clinicaltrials.gov / Patents & Registered Clinical Trials are quite different
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174149968&txt2find=Registered
Sources should be obvious.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174429360
and
USPTO
https://ppubs.uspto.gov/pubwebapp/static/pages/ppubsbasic.html
Page 1 of umpteen pages:
.png)
When did they file a PI/II trial? What information source are you referencing?
If they actually get a trial running and advancing for a change, how are they going to pay for it? Dilution at 20% off market rate (which has been trips for a while)?
Additionally, the Company has filed a Phase I/II clinical trial for COPD and possesses preclinical data for pulmonary fibrosis.
Onward.
Great another patent. How many do they have now? 7? How has that worked out for shareholders?
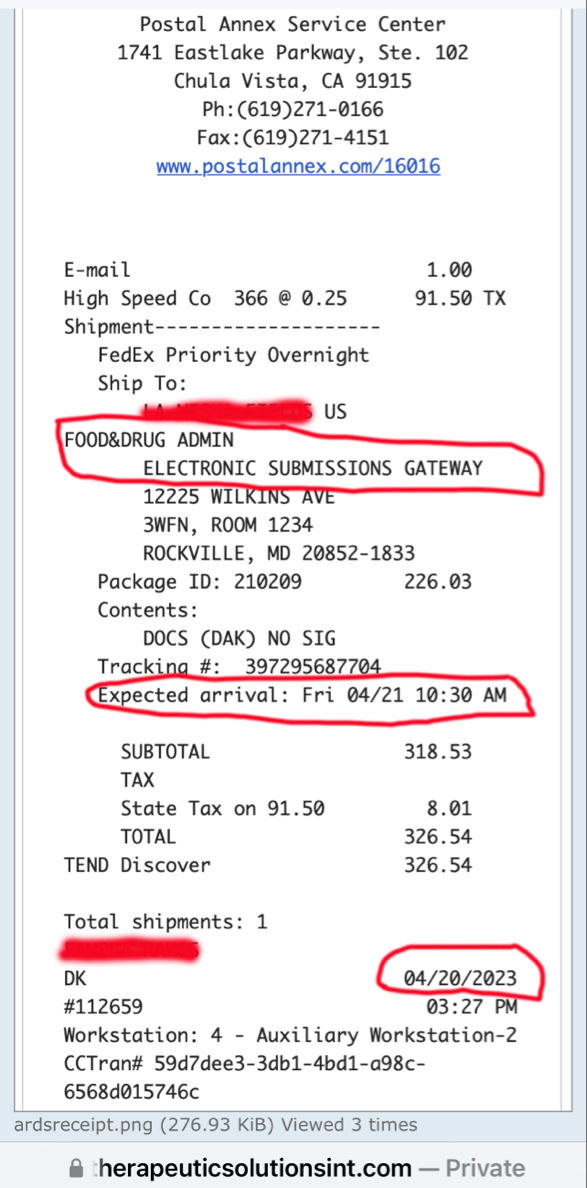
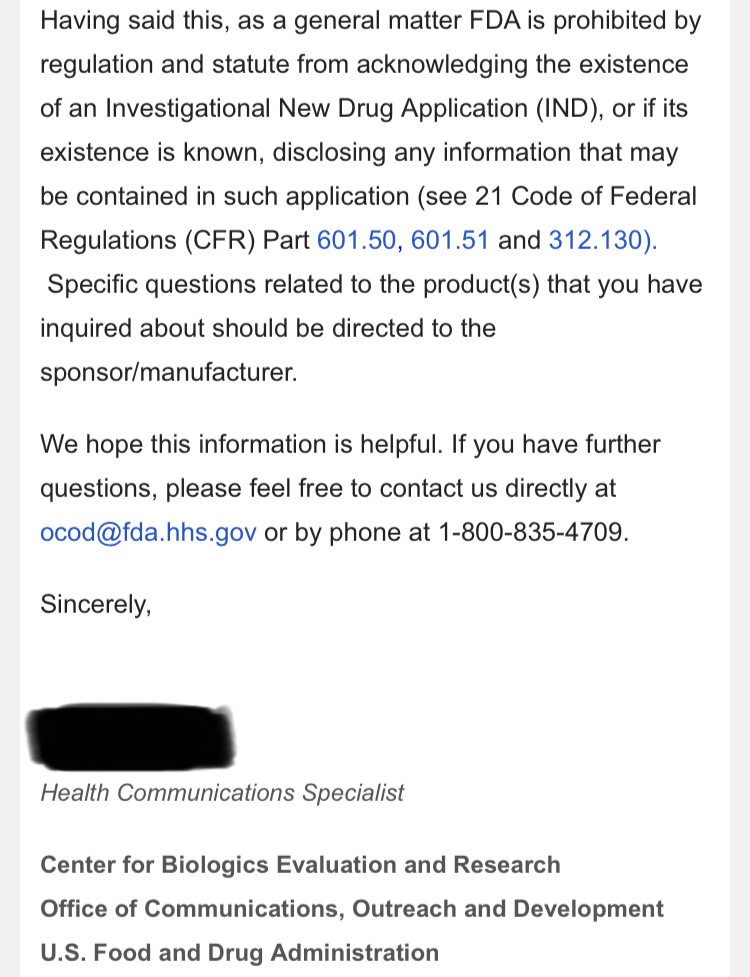
The press release claims that TSOI "owns a Phase III Investigational New Drug application for treatment of COVID-19." How does TSOI own any of that when it defaulted on the purchase agreement it had with the University of Miami (which means it does not own it)?
Working on a new application? The one that was submitted many months ago and went no where? What are they working on? A P1 application given that TSOI does not have a completed P2?
Who cares about Medistem? Many TSOI shareholders have lost more than 75% over course of the past year alone.
Therapeutic Solutions International Receives Notice of Allowance for Landmark Patent Covering Cellular Therapy of Lung Diseases
Clinical Stage Stem Cell Company Granted Exclusivity on the Use of Specific Immune System Cells to Increase Efficacy of Stem Cells in Lung Inflammation
ELK CITY, Idaho--(BUSINESS WIRE)-- Therapeutic Solutions International, Inc. (TSOI) announced today receipt of a Notice of Allowance from the United States Patent and Trademark Office regarding its patent application entitled “Stimulation of Mesenchymal Stem Cell Therapeutics Activities by Regulatory T Cells.”
The issued patent claims include:
“A method of inhibiting lung inflammation comprising: a) identifying a subject suffering from lung inflammation; b) administering a population of T regulatory cells in combination with a population of plastic adherent umbilical cord derived mesenchymal stem cells in an amount sufficient to reduce inflammation in said subject’s lungs.”
Therapeutic Solutions International owns a Phase III Investigational New Drug application for treatment of COVID-19. However, the Company is working on a new application for treating “all-cause” acute respiratory distress syndrome (ARDS) using the company’s patented JadiCell1, a true mesenchymal stem cell. Additionally, the Company has filed a Phase I/II clinical trial for COPD and possesses preclinical data for pulmonary fibrosis. All of these conditions are examples of lung inflammation.
“The claims issued today, in our opinion, capture the whole area of using a specific type of immune system cell, termed ‘T Regulatory Cells’ for enhancing activity of mesenchymal stem cells in all the major lung pathologies,” said Dr. Thomas Ichim, Board Member of TSOI and co-inventor of the patent. “We anticipate significant value in the current patent based on other companies working in the area of T regulatory cells. This is validated by the Regeneron-Sonoma Therapeutics2 and the Quell-AstraZeneca3 deals. It is nice to know that we own a potent piece of intellectual property that anyone hoping to enter this space will need to license.”
Dr. Ichim has a strong history of success in the area of biotechnology having taken his company Medistem from a 7 cent share price when he was made CEO to a sale at $1.35 to Intrexon, a NYSE traded company. Dr. Ichim’s lifetime of science, FDA clearances, and over 400 issued and published patents, have taken multiple companies to NASDAQ public listings.
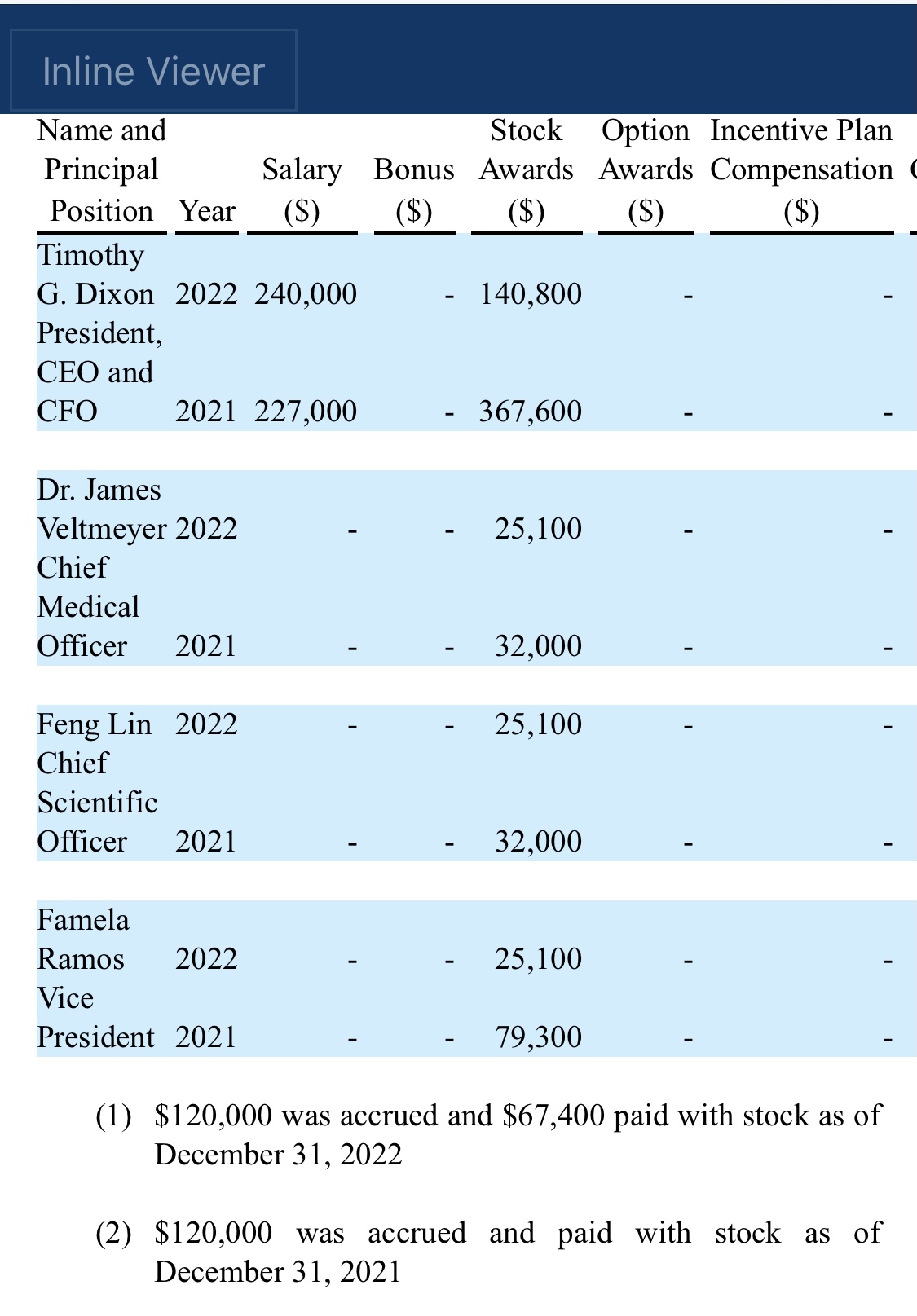
“At Therapeutic Solutions International we are building a critical mass of clinical data, scientific discoveries, and patents4,” said Timothy Dixon, President, and CEO of the Company and co-inventor. “We believe we are developing significant traction which will result in partnerships and co-development deals.”
About Therapeutic Solutions International, Inc.
Therapeutic Solutions International is focused on immune modulation for the treatment of several specific diseases. The Company's corporate website is www.therapeuticsolutionsint.com.
1 https://patents.google.com/patent/US9803176B2/en?oq=9803176
2 https://www.fiercebiotech.com/biotech/sonoma-biotherapeutics-takes-big-bite-120m-regeneron-collab-tacking-five-new-targets
3 https://www.reuters.com/business/healthcare-pharmaceuticals/astrazeneca-signs-2-bln-agreement-with-quell-develop-cell-therapies-2023-06-09/
4 https://patents.justia.com/assignee/therapeutic-solutions-international-inc
https://cts.businesswire.com/ct/CT?id=bwnews&sty=20240516930973r1&sid=acqr8&distro=nx&lang=en
View source version on businesswire.com: https://www.businesswire.com/news/home/20240516930973/en/
Timothy G. Dixon
ir@tsoimail.com
Source: Therapeutic Solutions International, Inc.
© Copyright Business Wire 2024
https://www.otcmarkets.com/stock/TSOI/news/story?e&id=2863154
$TSOI 8 x 9 w/ a 9 print & closes 00085 >(21.43%)
the subject quarterly report or transition report on Form 10-Q or subject distribution report on Form 10-D, or portion thereof, will be filed on or before the fifth calendar day following the prescribed due date;
Dixon knew & knows https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174296423&txt2find=ongoing
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174304345&txt2find=ongoing
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174258672&txt2find=ongoing
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174159413
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174149968&txt2find=ongoing
whose been responsible for TSOI's Ongoing pps decline & it's T/A Verified as well
Whew, I was wondering where everyone went!
And just like that CEO Tim Dixon called his own felony. Brilliant.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173001501
Trade Data since last T/A verified SS Update
Date Close/Last Volume Open High Low
05/09/2024 $0.0006 150,742,100 $0.0005 $0.0007 $0.0005
05/08/2024 $0.0006 170,815,400 $0.0007 $0.0007 $0.0005
05/07/2024 $0.0007 67,242,310 $0.0008 $0.001 $0.0007
05/06/2024 $0.0008 75,632,240 $0.0006 $0.0008 $0.0006
05/03/2024 $0.0006 57,890,520 $0.0005 $0.0006 $0.0005
05/02/2024 $0.0006 10,744,930 $0.0006 $0.0006 $0.0005
05/01/2024 $0.0006 17,697,640 $0.0005 $0.0006 $0.0005
04/30/2024 $0.0005 111,088,900 $0.0006 $0.0006 $0.0004
04/29/2024 $0.0006 48,979,020 $0.0007 $0.0007 $0.0005
04/26/2024 $0.0006 52,436,990 $0.0006 $0.0007 $0.0006
04/25/2024 $0.0006 22,166,110 $0.0006 $0.0006 $0.0005
04/24/2024 $0.0006 49,003,790 $0.0007 $0.0007 $0.0005
04/23/2024 $0.0006 25,939,830 $0.0007 $0.0007 $0.0006
04/22/2024 $0.0007 23,903,370 $0.0006 $0.0007 $0.0006
Authorized Shares 6,500,000,000 04/22/2024
Outstanding Shares 4,396,378,735 04/22/2024 >166,261,866
Restricted 1,454,564,260 04/22/2024 >57,857,142
Unrestricted 2,941,814,475 04/22/2024 >108,404,724
Held at DTC 2,839,780,106 04/22/2024 >56,818,182
T/A verifies Tim Dixon is responsible for the Ongoing pps decline
Authorized Shares 6,500,000,000 04/15/2024
Outstanding Shares 4,230,116,869 04/15/2024
Restricted 1,396,707,118 04/15/2024
Unrestricted 2,833,409,751 04/15/2024
Held at DTC 2,782,961,924 04/15/2024
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174251871
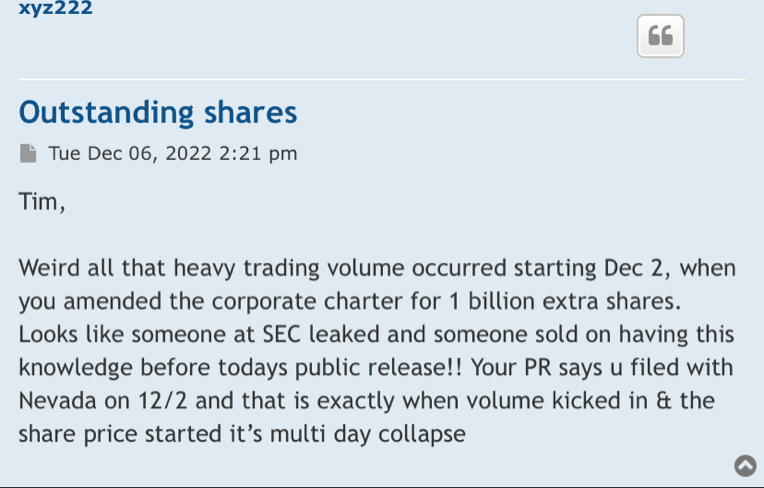
More than 320 Million in volume the past 2 days alone and the pps has dropped nearly 30%. Not a good look.
How much of that is dilution (which will continue to be TSOI's main funding source for the foreseeable future)? I am looking forward to the updated OS count. It looks that 2.5 BILLION shares per year dilution pace is continuing to hold if not accelerate.
Worth repeating.
Vol 150,742,100 (FWIW, #4 on iHub's Most-Active OTC List)
Buy Vol 114,644,287
Sell Vol 33,453,463
? Vol 2,644,350
0.000556 47,363,917 avg. 15:43:02 T/A verifies Discount'em Dixon is responsible for $TSOI's Ongoing pps decline
Transfer Agent Verified
Share data displayed on www.otcmarkets.com alongside a "Transfer Agent Verified" logo has recently been verified by the company’s transfer agent as reliable and trustworthy. This data is also disseminated through OTC Markets Group's market data feeds to investors and broker-dealers.
The Transfer Agent Verified Shares Program provides investors current and reliable share data. The program enables stock transfer agents to report their clients' share data, including authorized and outstanding shares, to OTC Markets Group on a regular basis via a secure, electronic file transfer.
In regards to your last Image:
|
Followers
|
529
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
64360
|
|
Created
|
10/04/08
|
Type
|
Free
|
| Moderators BigBadWolf johnnytrader33 JMC$ Yooperman Hogwarts | |||













































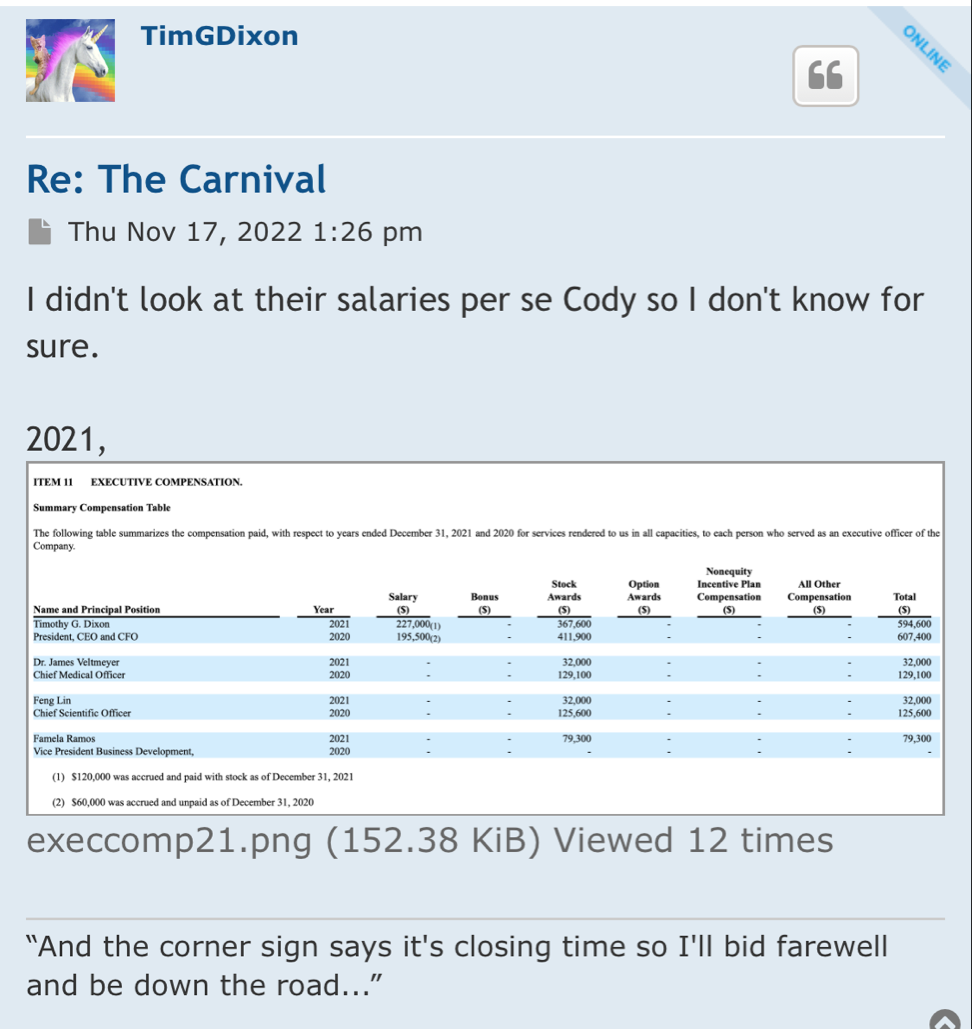


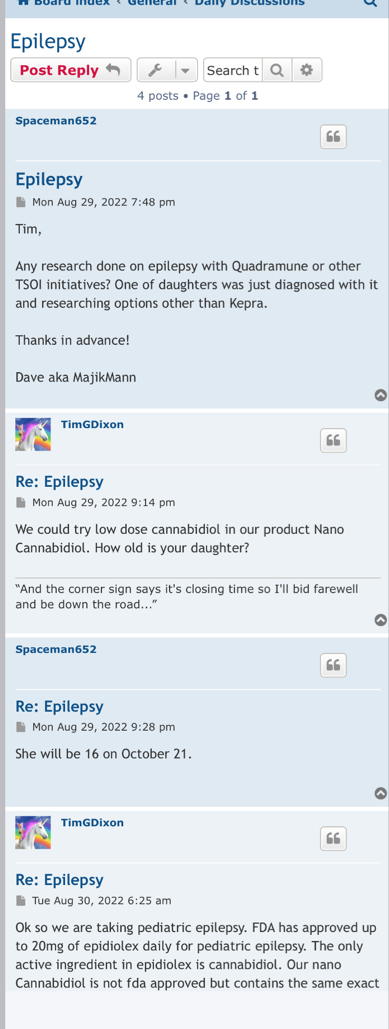
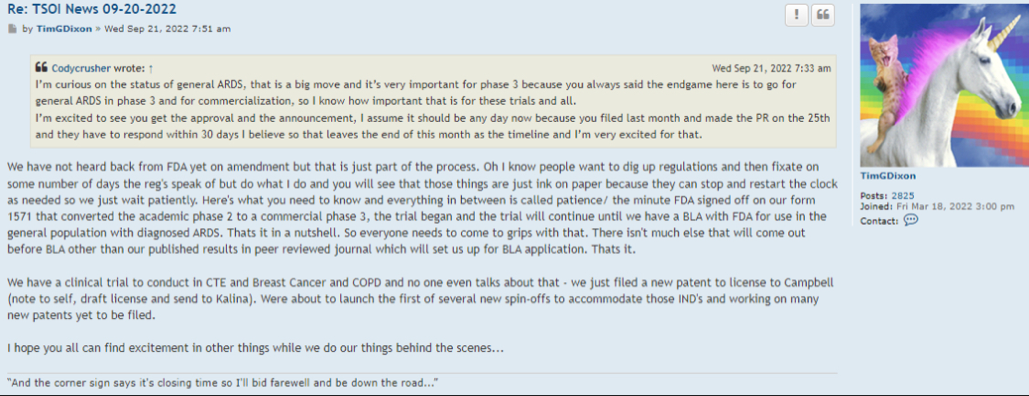








Preclinical Data Suggests QuadraMune™ Prevents Stress-Induced Suppression of Neurogenesis More Effectively than Prozac
OCEANSIDE, Calif., Dec. 9, 2020 /PRNewswire/ -- Therapeutics Solution International, Inc., (OTC Markets: TSOI), announced today new data suggesting the possibility that QuadraMune™ may mediate neuroprotective activity through preserving the ability of regenerative brain cells to proliferate subsequent to psychological stress.
The experiments, which involved exposing mice to established stressors, demonstrated that specific areas of the brain associated with production of new brain cells are damaged by stress. In agreement with previously published research, administration of fluoxetine (Prozac™) protected the brain from stress-induced damage. Surprisingly, QuadraMune™ administration appeared superior to Prozac™ at stimulating proliferation of new brain cells.
"QuadraMune™ which is currently in a clinical trial for prevention of COVD-191, has also been demonstrated to possess anti-inflammatory activity in other clinical trials, suppressing cytokines such as IL-62, which are known to be involved in depression3 and suicide4" said Kalina O'Connor, Director of Campbell Neurosciences and co-inventor on the patent. "Given major depressive disorder causes a significant risk for suicide, we are highly interested in exploring the use of QuadraMune™ for preventing suicide."
"Although much enthusiasm has been generated over the planned distribution of the COVID vaccine, at present little is being done to address mental health issues that are being exacerbated by the current pandemic" said Dr. James Veltmeyer, co-inventor of the patent, and Chief Medical Officer of the Company. "If current results are reproducible, the possibility that a nutraceutical would concurrently boost immunity while preserving mental health is highly enticing."
"It has not escaped us that COVID-19 is associated with increased inflammatory cytokines in the blood of patients, cytokines that also predispose to depression" said Famela Ramos, Vice President of Business Development for the Company. "It may be that the recent increase in suicides and suicide attempts is related biologically to activities of the coronavirus. It will be interesting to examine whether QuadraMune™ may modify putative negative mental effects of the virus."
"An estimated 17.3 million adults in the United States had at least one major depressive episode. This number represented 7.1% of all U.S. adults" stated Timothy Dixon, President and CEO of the Company. "We believe the Mission of our Company is not just providing a return on investment to our shareholders, but also increasing the quality of life for Americans. We are extremely pleased to report this unexpected finding with significant potential implications to advancing non-toxic means of helping patients with this terrible condition."
1 QuadraMune(TM) for Prevention of COVID-19 - Full Text View - ClinicalTrials.gov
2 Therapeutic Solutions International Announces Positive Preclinical and Clinical Evaluation of Nutritional Supplement QuadraMune™, Designed to Protect Against COVID-19 | BioSpace
3 Ting et al. Role of Interleukin-6 in Depressive Disorder. Int J Mol Sci. 2020 Mar 22;21(6):2194.
4 O'Donovan et al. Suicidal ideation is associated with elevated inflammation in patients with major depressive disorder. Depress Anxiety. 2013 Apr;30(4):307-14.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
