Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Nice article today
Trading currently at 27 cents and with a market cap of $74 million, Jaguar Health (NASDAQ:JAGX) sits firmly inside the penny stock category. The company’s name gives a clue as to its mission, which is to use chemical derivatives from natural sources found in rainforests to develop novel pharmaceuticals.
These proprietary prescription medications are directed towards both people and animals with gastrointestinal issues such as an overactive bowel. Currently, one of the company’s subsidiaries, Napo Pharmaceuticals, is working to develop and commercialize a drug named crofelemer. This drug aims to treat chemotherapy-induced overactive bowel, which is a common symptom for many cancer patients.
With the drug currently in Phase 3 clinical trials, the potential for a spike in JAGX’s value is real. Though it will likely be a few more years, should the drug achieve regulatory approval, it could become a staple among chemotherapy treatments for symptom prevention. Pair this with aging populations around the developed world, and the demand for crofelemer could drive JAGX up even further
https://investorplace.com/2024/05/want-10000-invest-1000-in-these-3-penny-stocks-now/
Well that was the HUGE SCRE DROP DAY LOL NICE!!! Going .36+ this week!
Looks like we’re leveling off for the day. We’ll see what happens at power hour if it pops.
I will repeat myself, the SVP President international of ImmunoGen sold the company to ABBVIE earlier this year for 10 BILLION dollar.
That same guy has joined Jaguar as PRESIDENT of Jaguar International !!! OPEN YOUR EYES
Anyone not seeing the potential of this stock, doesn't deserve to be in it when the party starts.
Please leave or short it !
another one of those decision points. VOLUME has really dried up, guess is, holders, buyers and sellers are looking to see who moves 'first'..
I'm sure this is sykes and crew making their final exit.
Everyone is waiting for their results on Phase III trial for cancer therapy related diarrhea in humans (CTD): https://jaguar.health/pipeline/
Results should be out any day now, deadline is June, i think.
Name of the game I guess lol dead cat bounce we are not
chemo, but when? Is there enough to keep it going right now?
Damn if this isn’t a FU from buyers I don’t know what is!!! AWESOME recovery!
as is the 'norm' when the pullback first commences, 'some' see the shares as 'on discount/cheap'
and start nibbling..
Course, one never knows where it will land at till it does, so sometimes catching the 'falling knife' does work out..
Good luck..
disclaimer: just watching. Using 32-33 cents now as a 'target price' on the upside, tho I had thought 50 cents was possible
if they had released the PH 3 data while the momentum was high.
have to see where it bottoms out at and whether or not its worth grabbing another round of shares:
28 cents to 32 cents would be a 15% return... for example..
There's bigger catalyst here to make this Solidify.
Is dog diarrhea approval the catalyst here or something bigger coming, and when?
Love watching my position go down $2000 then right back up lol
Exactly hitting all the stop losses!!! Love hate this kinda play from them !!! Uhhh
MM’s are killing pps today. Evil bastards. Someone wants to buy a boat load cheap I think.
Just needs to stay above .26 below game over….
as fast as its run up, ya have to figure, those that jumped on board trying to get some, will leave just as fast once they realize the
party is over for the moment.
I use the Moving Average line as a 'target' for the share price to bounce of on a pullback. Its down around 23 cents this morn (up from 21 cents' the other
day).
As always, the best we can do is 'guess' and then see what happens, and if we are so inclined, be ready to take advantage of it.. (buying opportunity)..
Unfortunately, as essentially a penny stock, no real institutional support at this price level?? It could get just as nasty as it did 'good'..
Gonna be ugly today MMs going to shake the TREE!!!
Any opinions on this : The new president of Jaguar International , was (co-) responsible for the biggest buy out deal of 2023. They sold ImmunoGEN for 10 BILLION dollars to ABBVIE, after that ImmunoGen reported positive phase 3 data in May 2023, earlier that year.
Seems like that guy seems a similar opportunity with Jaguar Health? Maybe not in buy out deal, but in a partnership deal?
Don't think so ... already has gone up a bit....Flat to Down probably
Prescription drug could prevent cancer therapy-related diarrhoea
Stomach pain
30 January 2024
Posted in Cancer Research, News | Tagged Cancer treatment, Chemotherapy, Napo Pharmaceuticals, Oncology, Preclinical investigations, Puma Biotechnology
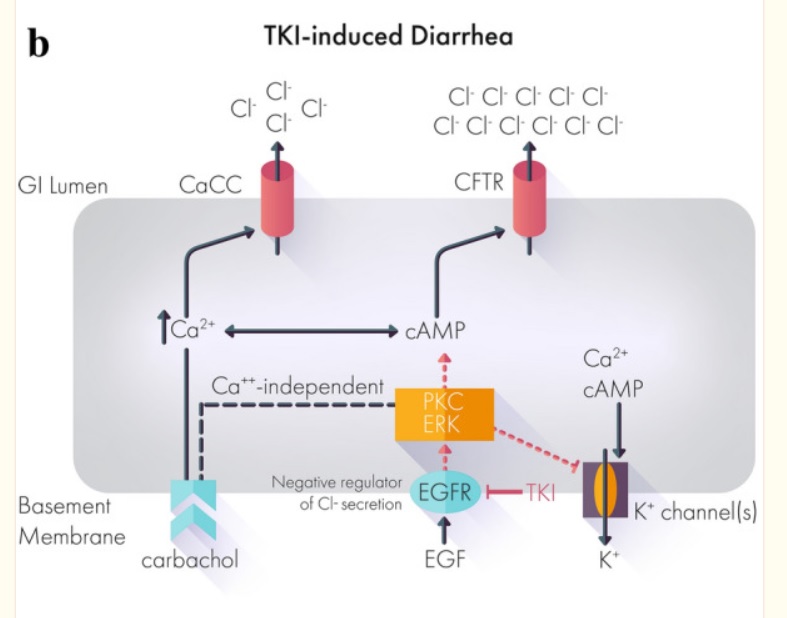
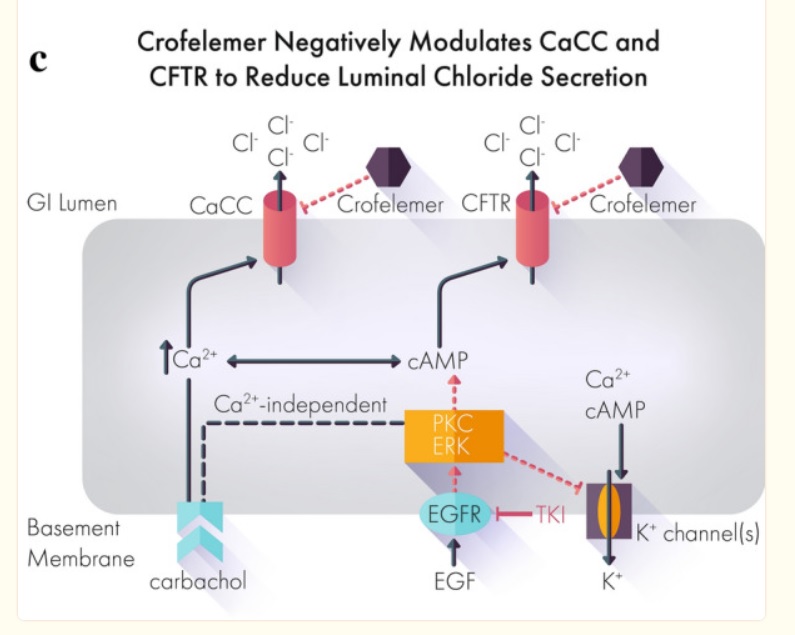
A study in dogs has shown that crofelemer can provide symptomatic relief of diarrhoea associated with the irreversible pan-human epidermal growth factor receptor (HER) tyrosine kinase inhibitor (TKI) neratinib (Nerlynx).
Puma Biotechnology’s Nerlynx is a pan HER TKI indicated for the extended adjuvant treatment of adult patients with early stage and metastatic HER2 positive breast cancer.
The study was conducted by Jaguar company Napo Pharmaceuticals in collaboration with Puma Biotechnology.
Cancer therapy-related diarrhoea (CTD) has the potential to cause dehydration, infections and non-adherence to treatment, and is a growing unmet medical need in cancer patients.
“We are very pleased with the results of this prophylactic study, which was undertaken to ‘model’ the human situation with neratinib,” said Lisa Conte, Jaguar’s President and CEO. “Crofelemer, our novel, oral, plant-based, non-opioid antidiarrhoeal prescription medication, is also the subject of OnTarget, Jaguar’s pivotal Phase III clinical trial for the follow-on indication of the preventative treatment of CTD, an indication we also refer to as chemotherapy-induced overactive bowel (CIOB).”
Dr Michael Guy, Jaguar’s Vice President of Preclinical and Nonclinical Studies, added: “The study demonstrated that crofelemer caused an approximate 30% reduction in the incidence and severity of diarrhoea associated with daily oral administration of neratinib, which was statistically significant.”
In a pilot Phase II study in humans, seven patients underwent treatment with crofelemer and loperamide, five of whom were able to control diarrhoea with crofelemer alone.
“Though this study involved a small number of patients, the results show activity of crofelemer for the management of neratinib-induced diarrhea, which warrants further investigation of crofelemer for treatment of cancer therapy-related diarrhea (CTD),” said study investigator Hope Rugo, Professor of Medicine and Winterhof Family Professor of Breast Oncology, University of California San Francisco Helen Diller Family Comprehensive Cancer Center.
Charts looks like a few down days then cup up Nike symbol uhhhh oh well I’m holding…
This study, conducted by Jaguar family company Napo Pharmaceuticals in collaboration with Puma Biotechnology, is one of three important studies providing scientific rationale for the use of crofelemer for prevention and treatment of CTD Studies published in peer-reviewed journals and continuum of datapoints highlight the neglected and growing need for treatment of one of the most common and debilitating side effects of patients undergoing cancer therapy SAN FRANCISCO, CA / ACCESSWIRE / January 25, 2024 /Jaguar Health, Inc.(NASDAQ:JAGX) today announced that a dog study evaluating the effects of crofelemer in providing symptomatic relief of diarrhea associated with the irreversible pan-human epidermal growth factor receptor (HER) tyrosine kinase inhibitor (TKI) neratinib (Nerlynx®) has been published in the peer-reviewed journal PLOS ONE. The study, titled Effects of orally administered crofelemer on the incidence and severity of neratinib-induced diarrhea in female dogs, was conducted by Jaguar family company Napo Pharmaceuticals in collaboration with Puma Biotechnology, Inc. and can be viewed by clicking here. "We are very pleased with the results of this prophylactic study, which was undertaken to ‘model' the human situation with neratinib," said Lisa Conte, Jaguar's president and CEO. "Crofelemer, our novel, oral, plant-based, non-opioid antidiarrheal prescription medication, is also the subject of OnTarget, Jaguar's pivotal Phase 3 clinical trial for the follow-on indication of the preventative treatment of cancer therapy-related diarrhea (CTD), an indication we also refer to as chemotherapy-induced overactive bowel (CIOB) - which includes symptoms such as unpredictable and/or chronic debilitating diarrhea and GI urgency. Diarrhea, which has the potential to cause dehydration, infections, and non-adherence to treatment in cancer patients, is a growing unmet medical need in the age of targeted cancer therapies." Conte added, "This neratinib study was the first of three studies over the past several years that provide scientific rationale for the use of crofelemer in humans for treatment of CTD. This continuum of datapoints highlights the neglected and growing need for prophylaxis of diarrhea in cancer patients. Prophylactic therapies exist to help prevent chemotherapy-induced nausea and vomiting (CINV) in cancer patients, but no antidiarrheal medications are currently approved for treatment of CTD. Interestingly, there is currently one available product for diarrhea in dogs undergoing cancer treatment: Canalevia®-CA1 (crofelemer delayed-release tablets), our FDA conditionally approved prescription drug for the treatment of chemotherapy-induced diarrhea (CID) in dogs." Effects of orally administered crofelemer on the incidence and severity of neratinib-induced diarrhea in female dogs "This 28-day preclinical pharmacological study in healthy female dogs was designed to evaluate the scientific rationale for the use of crofelemer in humans in reducing the severity and incidence of diarrhea associated with a tyrosine kinase inhibitor (neratinib)," said Dr. Michael Guy, D.V.M., M.S., Ph.D., Jaguar's vice president of preclinical and nonclinical studies. "The study was conducted without the prophylaxis or concomitant use of loperamide and demonstrated that crofelemer caused an approximate 30% reduction in the incidence and severity of diarrhea associated with daily oral administration of neratinib, which was statistically significant. Crofelemer also demonstrated significant improvement in the proportion of responder dogs, and there was a trend for fewer neratinib dose reductions in both crofelemer treatment groups when compared to the control group." Nerlynx, a drug product of Puma Biotechnology, Inc., is a pan HER TKI indicated for the extended adjuvant treatment of adult patients with early stage HER2 positive breast cancer and also for metastatic HER2 positive breast cancer. "Diarrhea was reported in 95% of Nerlynx-treated patients in ExteNET, a randomized placebo-controlled trial in the extended adjuvant setting who were not required to receive antidiarrheal prophylaxis," said Dr. Guy. "Nerlynx dose reductions due to diarrhea are often needed, and patients are advised to initiate antidiarrheal prophylaxis. Unlike crofelemer, which is already approved for a chronic use, synthetic antidiarrheal antimotility drugs are opioids and can bring risks in higher doses, often lead to constipation, and are thus not suitable for long-term use." Crofelemer for the Management of Neratinib-Associated Diarrhea in Patients With HER2+ Early-Stage Breast Cancer This independent pilot phase 2 study, which was published in the peer reviewed journal Clinical Breast Cancer in October 2023, can be viewed by clicking here. The study results indicate that crofelemer may be effective for the management of neratinib-induced diarrhea. "Seven patients underwent treatment with crofelemer and loperamide in this study, 5 of whom were able to control diarrhea with crofelemer alone. Though this study involved a small number of patients, the results show activity of crofelemer for the management of neratinib-induced diarrhea, which warrants further investigation of crofelemer for treatment of cancer therapy-related diarrhea (CTD)," said study investigator Hope S. Rugo, MD, Professor of Medicine and Winterhof Family Professor of Breast Oncology, University of California San Francisco Helen Diller Family Comprehensive Cancer Center. Dr. Rugo is a member of Jaguar's Scientific Advisory Board. This single center, open label trial enrolled patients with Stage 2 to 3 HER2+ breast cancer receiving neratinib in the extended adjuvant setting. One cohort took prophylactic crofelemer 125 mg BID and loperamide in the first 2 cycles, and as needed in the subsequent cycles. The second cohort received escalating doses of neratinib with loperamide. The primary endpoint was incidence of grade ≥3 diarrhea in the first 2 cycles. HALT-D: A Randomized Open-Label Phase II Study of Crofelemer for the Prevention of Chemotherapy-Induced Diarrhea in Patients with HER2-Positive Breast Cancer Receiving Trastuzumab, Pertuzumab, and a Taxane This investigator-initiated study, which was published in the peer reviewed journal Breast Cancer Research and Treatment in October 2022 and funded with support from Genentech, Inc., a member of the Roche group, can be viewed by clicking here. The targeted cancer therapies evaluated in the study cause diarrhea in up to 80% of breast cancer patients, reaching grade 3, which often requires hospitalization, in 8-12% of patients. "Diarrhea is a debilitating side effect that many people experience when undergoing breast cancer treatment that demands research attention," said the study's senior investigator, Sandra M. Swain, MD, professor of medicine at Georgetown University and a member of its Lombardi Comprehensive Cancer Center. "This is one of few studies to explore ways of proactively preventing diarrhea in women undergoing lifesaving treatment for their breast cancer. While we have more work to do, this study revealed important information about the treatment of diarrhea in this particular population." The results of the HALT-D study were released previously by the investigators at the 2021 San Antonio Breast Cancer Symposium by lead study investigator Paula Pohlmann, MD, PhD, formerly from Georgetown University and now Associate Professor, MD Anderson Cancer Center. HALT-D included 51 breast cancer patients scheduled to receive at least three cycles of chemotherapy and randomly assigned to either crofelemer in cycles 1 and 2 or the control group, which received standard of care. Antidiarrheal medications for breakthrough diarrhea were permitted but not given prophylactically. REMINDER: Multinational Association of Supportive Care in Cancer (MASCC) Webinar on January 29, 2024 A Multinational Association of Supportive Care in Cancer (MASCC) webinar titled "GI Toxicities from Cancer Therapies - Prevention and Management" is taking place Monday, January 29, 2024 from 11:30 AM to 12:30 PM Eastern Standard Time. The webinar was made possible by an unrestricted educational grant from Jaguar family company Napo Pharmaceuticals. To register for the webinar, click here. MASCC does not endorse or promote products or services of any industry partner including Jaguar Health, Inc. About Cancer Therapy-Related Diarrhea (CTD) More than 1.9 million people were expected to be diagnosed with cancer in 2023 in the U.S., according to the American Cancer Society. A significant proportion of patients undergoing cancer therapy experience diarrhea, and diarrhea has the potential to cause dehydration, infections, and non-adherence to treatment in this population. Novel "targeted cancer therapy" agents, such as epidermal growth factor receptor (EGFR) antibodies and tyrosine kinase inhibitors (TKIs), with or without cycle chemotherapy agents, may cause increased electrolyte and fluid content in the gut lumen, which results in passage of loose/watery stools (i.e., diarrhea). Diarrhea has been reported as one of the most common side effects of TKIs and may result in cancer therapy drug holidays or reductions from therapeutic dose, potentially impacting patient outcome. Diarrhea is also a common side effect of some approved CDK 4/6 inhibitors. Patients with CTD are 40% more likely to discontinue their chemotherapy or targeted therapy than patients without CTD,1 and the cost of care of CTD patients is estimated to be 2.9 times higher than for patients who are not experiencing CTD.2 About the Jaguar Health Family of Companies Jaguar Health, Inc. (Jaguar) is a commercial stage pharmaceuticals company focused on developing novel proprietary prescription medicines sustainably derived from plants from rainforest areas for people and animals with gastrointestinal distress, specifically associated with overactive bowel, which includes symptoms such as chronic debilitating diarrhea, urgency, bowel incontinence, and cramping pain. Jaguar family company Napo Pharmaceuticals focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states. Napo Pharmaceuticals' crofelemer drug product candidate is the subject of the OnTarget study, a pivotal Phase 3 clinical trial for preventive treatment of chemotherapy-induced overactive bowel (CIOB) in adults with cancer on targeted therapy. Jaguar family company Napo Therapeutics is an Italian corporation Jaguar established in Milan, Italy in 2021 focused on expanding crofelemer access in Europe and specifically for orphan and/or rare diseases. Jaguar Animal Health is a Jaguar tradename. Magdalena Biosciences, a joint venture formed by Jaguar and Filament Health Corp. that emerged from Jaguar's Entheogen Therapeutics Initiative (ETI), is focused on developing novel prescription medicines derived from plants for mental health indications.
Read more at: https://www.miamiherald.com/press-releases/article284671171.html#storylink=cpy
This is the BIG PR WE ARE ALL WAITING FOR… NOT THE DOG ONE….
JAGX the company is awaiting results from its Good Clinical Practice vendors supporting the trial. The results are “forthcoming,” the San Francisco-based company said. “The OnTarget trial is our paramount near-term clinical milestone,” said Chief Executive Lisa Conte. “We look forward to announcing and presenting the top line results when they are available
it's on their website, if you're a serious investor I suggest signing up for their email list. they will send you all updates :)
https://jaguarhealth.gcs-web.com/
Just keeping it real been daytrading 20 years hype is fun but once it dries up move on…. Plenty of opportunities
Tahoe
your comment on the potential market is spot on. Just how many DOGS need diarrhea medicine can there be?? What will it sell for,? Profit margin
all come to mind as questions.
Why I said my biggest concern is with whats a justifiable target price even if they get the PH3 results approved by the fda..
That said tho, when it comes to momentum on PR, there is no way to judge how long a 'run' will last, how high the share price goes, till its over..
Good luck..
Disclaimer: still observing the 'skirmish' taking place for direction. Who knows, maybe it will consolidate at the price range of 30-32 cents.. That would be nice to
see.. Would show nice support...
But let's do .48 first. Then pull back.....
Looking on the daily, i still think it can revisit ~.26 before going higher, a healthy pullback. If no news the following days, it could do this, imo.
The issue is that there are already a bunch of old school meds that are very effective and also very cheep.
So…. If marketing just towards chemotherapy patients, pretty small market in animals.
From my Vet friend
still looking for direction.. Volume appears to be tapering off in comparison to the prior 2 days of heavy volume as it rallied up.
Now appears that 'both' sides are wondering who 'blinks' first..
Breaks .33 it’s off to the races again 10% up days I’m cool with slow and steady not rocketship and dump
Told everyone over at MOMO group maybe they jumping in now
Shot just go green Positive already! the surge will come this afternoon
Hoping it goes over .40 this week. That would be sweet. Jags should be able to push over a dollar without a split.
Looks like the heard jumped into other 20%+ bangers today… AGBA MULN SOUN.. oh well they will be back in… GMZP if you wanna play that kinda game. JAGX is my highest position right now with 50,777 shares yes I love gambling lol
|
Followers
|
157
|
Posters
|
|
|
Posts (Today)
|
1
|
Posts (Total)
|
6338
|
|
Created
|
12/01/15
|
Type
|
Free
|
| Moderators ProfitChaser oldstocks DTGoody powerbattles crudeoil24 SmellMyFinger | |||
Jaguar Health, Inc. $JAGX
Bringing Plant-based Medicines to Life™
Pharmaceutical Manufacturing
San Francisco, California
Jaguar Health’s organizational framework is designed to enable our product pipeline to reach the world’s interconnected marketplace, allowing for development and commercialization opportunities worldwide.
Products:

Mytesi Crofelemer 125mg delayed release tablets
Mytesi is FDA-approved for symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy
Market size of the FDA HIV Mytesi Crofelemer could be $25-$30 Million in the USA
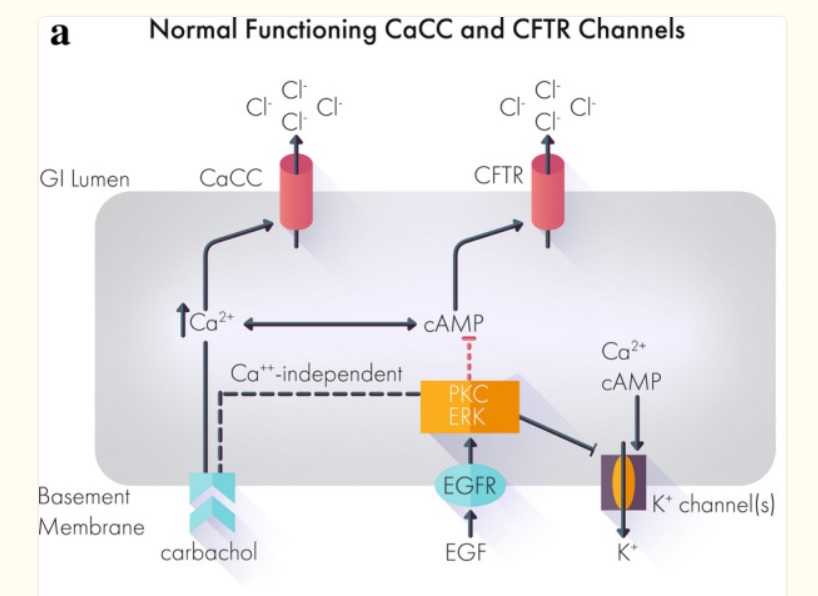
Mytesi has a novel mechanism of action that modulates chloride secretion in the gastrointestinal tract, reducing excessive chloride and accompanying water flow and relieving watery diarrhea.
Mytesi is not indicated for the treatment of infectious diarrhea. In clinical studies, the most common adverse reactions occurring at a rate greater than placebo were upper respiratory tract infection (6%), bronchitis (4%), cough (4%), flatulence (3%), and increased bilirubin (3%).
Canalevia-CA1 Crofelemer delayed release tablets
Distributors listed:
https://www.canalevia.com/distributor-information
CANALEVIA-CA1 is conditionally approved for the treatment of chemotherapy-induced diarrhea (CID) in dogs.
Conditionally approved by FDA pending a full demonstration of effectiveness under application number 141-552.
CAUTION: Federal (USA) law restricts this drug to use by or on the order of a licensed veterinarian. It is a violation of Federal Law to use this product other than as directed in the labeling.

Lytham Partners Investor Select Conference February 1st, 2024
https://wsw.com/webcast/lytham10/jagx/2084640
Pipeline: https://jaguar.health/pipeline/
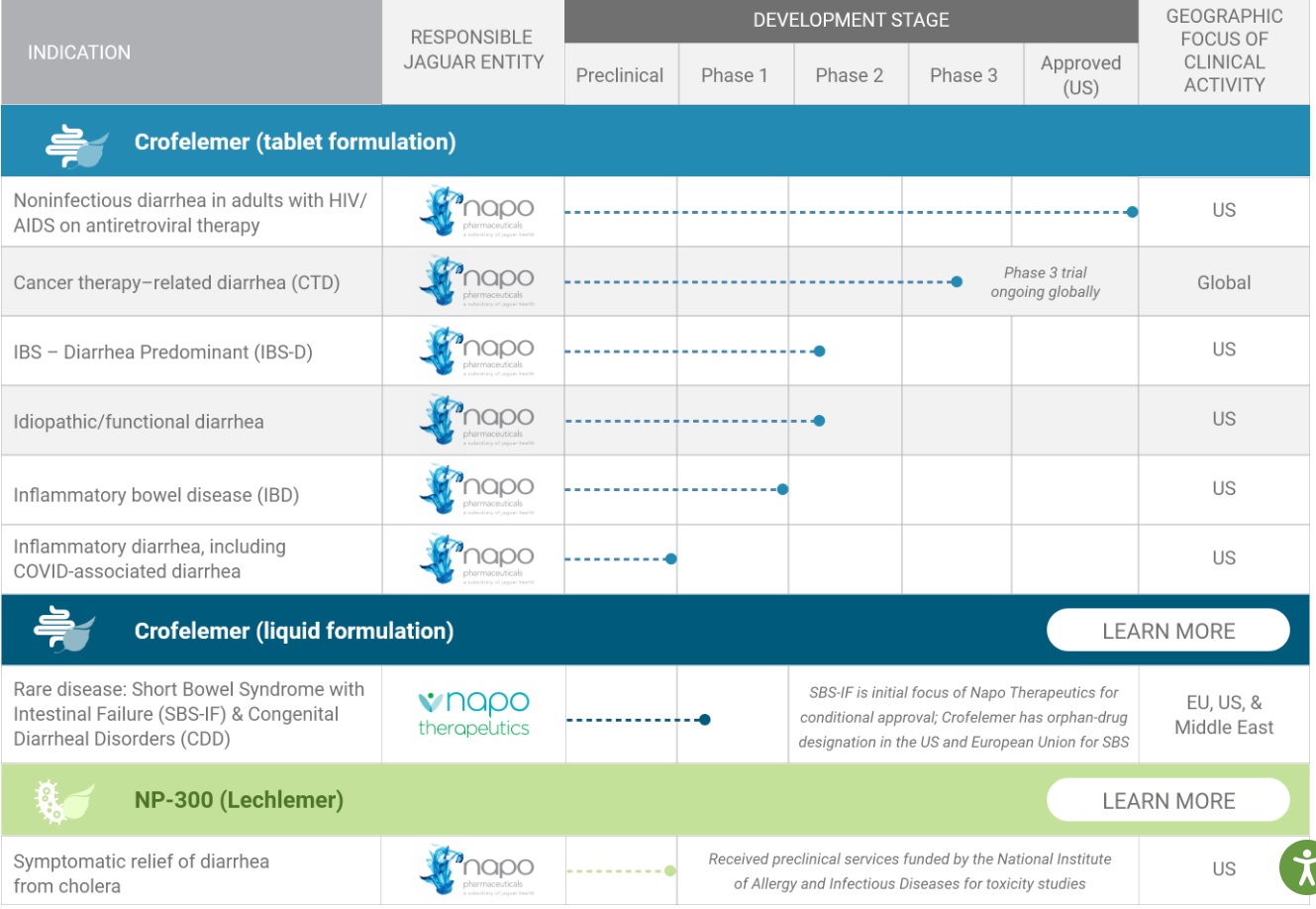
How to calculate the value of drugs and biotech companies
https://www.baybridgebio.com/drug_valuation.html
About the Jaguar Health Family of Companies
Jaguar Health, Inc. (Jaguar) is a commercial stage pharmaceuticals company focused on developing novel proprietary prescription medicines sustainably derived from plants from rainforest areas for people and animals with gastrointestinal distress, specifically associated with overactive bowel, which includes symptoms such as chronic debilitating diarrhea, urgency, and bowel incontinence.
Jaguar family company Napo Pharmaceuticals focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states.
Napo Pharmaceuticals' crofelemer drug product candidate is the subject of the OnTarget study, a pivotal Phase 3 clinical trial for preventive treatment of chemotherapy-induced overactive bowel (CIOB) in adults with cancer on targeted therapy.
Jaguar family company Napo Pharmaceuticals focuses on developing and commercializing human prescription pharmaceuticals for essential supportive care and management of neglected gastrointestinal symptoms across multiple complicated disease states.
Napo Pharmaceuticals' crofelemer drug product candidate is the subject of the OnTarget study , a pivotal Phase 3 clinical trial for preventive treatment of chemotherapy-induced overactive bowel (CIOB) in adults with cancer on targeted therapy. Jaguar family company Napo Therapeutics is an Italian corporation Jaguar established in Milan, Italy in 2021 focused on expanding crofelemer access in Europe and specifically for orphan and/or rare diseases. Jaguar Animal Health is a Jaguar tradename. Magdalena Biosciences, a joint venture formed by Jaguar and Filament Health Corp. that emerged from Jaguar's Entheogen Therapeutics Initiative is focused on developing novel prescription medicines derived from plants for mental health indications.
A Case Report: Improved Control of Tyrosine Kinase Inhibitor-Induced Diarrhea
Despite the efficacy of tyrosine kinase inhibitors (TKIs) across multiple cancers, side effects including treatment-related diarrhea can impede a patient’s ability to reach therapeutic doses or stay on therapy.
Below, we present the case of a 72-year-old patient with metastatic papillary renal cell carcinoma recurrent despite nephrectomy.
Over the course of treatment, the patient received multiple different tyrosine kinase inhibitors with varying efficacy.
Treatment with the TKI cabozantinib after failure of two prior TKIs resulted in a clinical response with shrinkage of his nodal metastatic disease.
However, the severe treatment-related diarrhea refractory to conventional management required both dose holds and dose reductions of cabozantinib.
Off-label administration of crofelemer, a novel FDA-approved antidiarrheal agent, successfully controlled the treatment-related diarrhea and allowed resumption and partial dose increase of cabozantinib.
This case suggests that crofelemer could be a viable therapeutic strategy to address TKI-induced diarrhea.
9.8 million people
receive chemotherapy worldwide per year
Wilson B, Jacob S, Yap M, et al. Estimates of global chemotherapy demands and corresponding physician workforce requirements for 2018 and 2040: a population-based study. Lancet Oncol. 2019;20(6):769-780. doi:10.1016/S1470-2045(19)30163-9
Chemotherapy-induced
diarrhea occurs in
50%-80%
of treated patients
Stein A, Voigt W, Jordan K. Chemotherapy-induced diarrhea: pathophysiology, frequency and guideline-based management. Ther Adv Med Oncol. 2010;2(1):51-63. doi:10.1177/1758834009355164
Patients with cancer therapy–
related diarrhea (CTD) were
40% more likely to
discontinue chemotherapy or
targeted cancer therapy than
patients without CTD
Okhuysen PC, Schwartzberg LS, Roeland E, et al. The impact of cancer-related diarrhea on changes in cancer therapy patterns. J Clin Oncol. 2021;39(15):12111. doi:10.1200/JCO.2021.39.15_suppl.12111
Research & Development
In October 2020, Napo Pharmaceuticals initiated a pivotal Phase 3 clinical trial
This Phase 3 study is evaluating crofelemer for prophylaxis of diarrhea in adult cancer patients receiving targeted therapy
The principal investigator and co-investigators identified are from MD Anderson Cancer Center
Jaguar Health https://jaguar.health/
Napo Pharmaceuticals https://napopharma.com/
Napo Therapeutics https://napotherapeutics.com/en/
https://www.magdalenabiosciences.com/
Magdalena is a joint venture formed by Jaguar Health and Filament Health. The company holds an exclusive license to Jaguar's proprietary library of approximately 2,300 highly characterized medicinal plants and 3,500 plant extracts for designated therapeutic indications.
Visit Jaguar on LinkedIn:
https://www.linkedin.com/company/jaguar-health/
Visit Jaguar on Instagram: https://www.instagram.com/jaguarhealthcommunity/
Visit Jaguar on X: https://twitter.com/Jaguar_Health
Phone: 415-371-8300
Contact: hello@jaguar.health
Press Releases: https://jaguarhealth.gcs-web.com/news-releases
Most if not all works by mechanism chloride ions into the gut then water comes and diarrhea out. Crofelemer normalizes abnormal ion flow targeting only locally in the gut.
Jaguar has been putting the expense into making sure Crofelemer drug supply is ready to get into this opportunity to treat cancer related chemo patients since pharmacy’s already can get FDA approved, all ready in the in the supply chain in the pharmacy as Crofelemer for AIDs patients.
Research has shown it costs 3x as much to treat a cancer patient with diarrhea.
It would be better to treat the cancer patient for diarrhea with Crofelemer pending OnTarget Study and FDA approval.
Since Crofelemer is FDA approved to treat dogs for Cancer Chemo related Diarrhea it has been said:
If your going to have cancer and cancer related Diarrhea, it’s better to be a dog than a human right now.
The companies that manufacture the cancer agents/drugs sort of point to the availability of Crofelemer because it allows them to keep their patients on their therapies thereby improving their penetration and their revenues opportunity.
3rd party market research put Short Bowel Syndrome at $5-$12 Billion opportunity because these patience are so ill, high mortality, high morbidity (the state of being unhealth for a particular disease or situation), so high of expense to manage through life.
That opportunity can be reached in small trials around the world to show benefits to be able to show global development.
Product already approved, already fully in the supply chain in the pharmacy.
Potential Market of Mytesi (crofelemer)
Jaguar and Napo $JAGX if the Phase 3 OnTarget Study has good results.
The Phase 3 OnTarget Study results are due to be out soon in this quarter.
The Phase 3 OnTarget study is testing whether the company's plant-based prescription drug crofelemer can prevent or substantially reduce diarrhea and other symptoms of CIOB when any one of the selected 24 different targeted therapies that are associated with diarrhea in patients is initiated.
Each year, more than 1 million cancer patients in the United States receive chemotherapy or radiation. Globally 9.8 million require chemotherapy.
Diarrhea is a common side effect of targeted therapies. Up to 95% of patients on tyrosine kinase inhibitors (TKIs) suffer from diarrhea, depending on the TKI used.
Any drug used to treat cancer (including tyrosine kinase inhibitors or TKIs) can be considered chemo, but here chemo is used to mean treatment with conventional cytotoxic (cell-killing) drugs that mainly kill cells that are growing and dividing rapidly. Chemo was once one of the main treatments for CML.
The cost for Mytesi oral delayed release tablet 125 mg is around $2,600 for a supply of 60 tablets. This amount is the one month supply as testing in the OnTarget Study.
That is a 24 week study = 5.6 month supply needed = $14,560.00 for treatment.
Potential Market of Mytesi (crofelemer)
100,000 patients a year = $1,456,000,000
250,000 patients a year = $3,640,000,000
500,000 patients a year = $7,280,000,000
1,000,000 patients a year = $14,560,000,000 (assuming worldwide)
5,000,000 patients a year = $72,800,000,000 (assuming worldwide)
The estimate of patients depends on how many quit taking chemo, the onTarget Study, FDA approval and or quit taking Mytesi (crofelemer).
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
